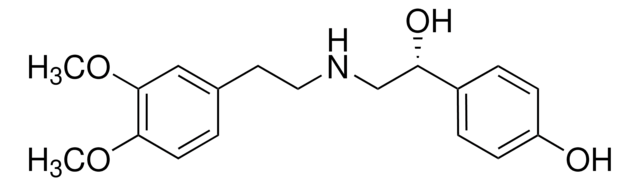
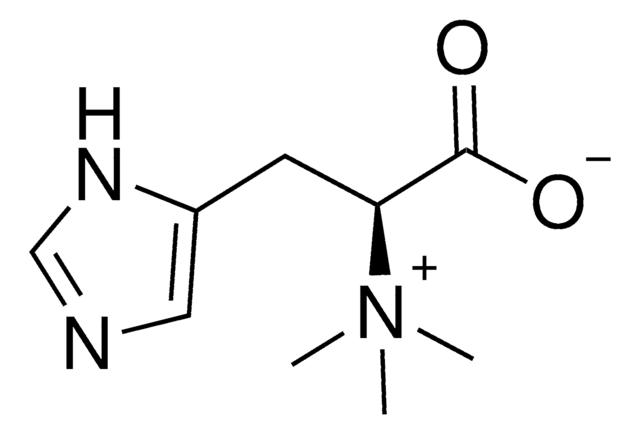
E7521
L-(+)-Ergothioneine
Synonyme(s) :
2-mercaptohistidine trimethyl betaine, Ergothioneine, Sympectothion, Thiasine, Thiolhistidine-betaine, Thioneine, (S)-α-Carboxy-N,N,N-trimethyl-2-mercapto-1H-imidazole-4-ethanaminium inner salt
About This Item
Produits recommandés
Source biologique
fungus (Actinomycetales)
fungus (Ascomycota)
fungus (Basidiomycota)
Niveau de qualité
Essai
≥98.0%
Forme
powder
Poids mol.
229.30
Conditions de stockage
(Keep container tightly closed in a dry and well-ventilated place)
Technique(s)
protein quantification: suitable
Solubilité
water: 50 mg/mL, clear, colorless
Température de stockage
−20°C
Chaîne SMILES
C[N+](C)(C)[C@@H](Cc1c[nH]c(S)n1)C([O-])=O
InChI
1S/C9H15N3O2S/c1-12(2,3)7(8(13)14)4-6-5-10-9(15)11-6/h5,7H,4H2,1-3H3,(H2-,10,11,13,14,15)/t7-/m0/s1
Clé InChI
SSISHJJTAXXQAX-ZETCQYMHSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Research area: Apoptosis
Application
L-(+)-Ergothioneine has been used:
- as a component of the maturation medium for cumulus-oocyte complexes (COCs) to test protective function on lipid peroxide formation
- as an antioxidant compound to test type 2 diabetes patients
- as a positive control in solute carrier protein 22 A4 (SLC22A4) transport assay
Actions biochimiques/physiologiques
Conditionnement
Autres remarques
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique