Alle Fotos(1)
Wichtige Dokumente
930431
SuTEx1-alkyne
≥95%
Synonym(e):
N-2-propyn-1-yl-4-(1H-1,2,4-triazol-1-ylsulfonyl)-benzamide
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C12H10N4O3S
CAS-Nummer:
Molekulargewicht:
290.30
MDL-Nummer:
UNSPSC-Code:
12352101
NACRES:
NA.21
Empfohlene Produkte
Beschreibung
Application: Chemoproteomics
Qualitätsniveau
Assay
≥95%
Form
powder
mp (Schmelzpunkt)
155 °C
Lagertemp.
−20°C
SMILES String
O=C(C1=CC=C(S(N2N=C([H])N=C2)(=O)=O)C=C1)NCC#C
InChIKey
ZBNCPAXYEUCPRL-UHFFFAOYSA-N
Anwendung
SuTEx1-alkyne is a probe that uses sulfur-triazole exchange chemistry to label tyrosines. A method was developed using cysteine-reactive compounds including this one to allow for unbiased analysis of proteomic data in quantitative applications (Zanon et al. 2021). The method uses light or heavy labelling with the isotopically labelled desthiobiotin azide (isoDTB) tag for mass spectrometry analysis (Zanon et al. 2020). Analysis then uses the isotopic tandem orthogonal proteolysis activity-based protein profiling (isoTOP-ABPP) workflow (Weerapana et al. 2010, Backus et al. 2016)
Sonstige Hinweise
Profiling the proteome-wide selectivity of diverse electrophiles
A quantitative thiol reactivity profiling platform to analyze redox and electrophile reactive cysteine proteomes
Ethynylation of Cysteine Residues: From Peptides to Proteins in Vitro and in Living Cells
A Chemoproteomic Platform To Assess Bioactivation Potential of Drugs
Inhibition of Zinc-Dependent Histone Deacetylases with a Chemically Triggered Electrophile
Reversibility of Covalent Electrophile-Protein Adducts and Chemical Toxicity
A quantitative thiol reactivity profiling platform to analyze redox and electrophile reactive cysteine proteomes
Ethynylation of Cysteine Residues: From Peptides to Proteins in Vitro and in Living Cells
A Chemoproteomic Platform To Assess Bioactivation Potential of Drugs
Inhibition of Zinc-Dependent Histone Deacetylases with a Chemically Triggered Electrophile
Reversibility of Covalent Electrophile-Protein Adducts and Chemical Toxicity
Ähnliches Produkt
Produkt-Nr.
Beschreibung
Preisangaben
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
Lot/Batch Number
Leider sind derzeit keine COAs für dieses Produkt online verfügbar.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Profiling the proteome-wide selectivity of diverse electrophiles
Patrick R. A. Zanon ,Fengchao Yu, et al
ChemRxiv : the preprint server for chemistry (2021)
Zarko V Boskovic et al.
ACS chemical biology, 11(7), 1844-1851 (2016-04-12)
Unbiased binding assays involving small-molecule microarrays were used to identify compounds that display unique patterns of selectivity among members of the zinc-dependent histone deacetylase family of enzymes. A novel, hydroxyquinoline-containing compound, BRD4354, was shown to preferentially inhibit activity of HDAC5
De Lin et al.
Chemical research in toxicology, 21(12), 2361-2369 (2009-06-24)
The biotin-tagged electrophiles 1-biotinamido-4-(4'-[maleimidoethylcyclohexane]-carboxamido)butane (BMCC) and N-iodoacetyl-N-biotinylhexylenediamine (IAB) have been used as model electrophile probes in complex proteomes to identify protein targets associated with chemical toxicity. Whereas IAB activates stress signaling and apoptosis in HEK293 cells, BMCC does not. Cysteine
A H El-Khatib et al.
Journal of mass spectrometry : JMS, 52(8), 543-549 (2017-06-04)
1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) derivatives are applied in quantitative proteomics owing to their ability to react with different functional groups, to harbor lanthanoides and hence their compatibility with molecular and elemental mass spectrometry. The new DOTA derivatives, namely Ln-MeCAT-Click and Ln-DOTA-Dimedone
Rui Sun et al.
Chemical research in toxicology, 30(10), 1797-1803 (2017-09-30)
Reactive metabolites (RM) formed from bioactivation of drugs can covalently modify liver proteins and cause mechanism-based inactivation of major cytochrome P450 (CYP450) enzymes. Risk of bioactivation of a test compound is routinely examined as part of lead optimization efforts in
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.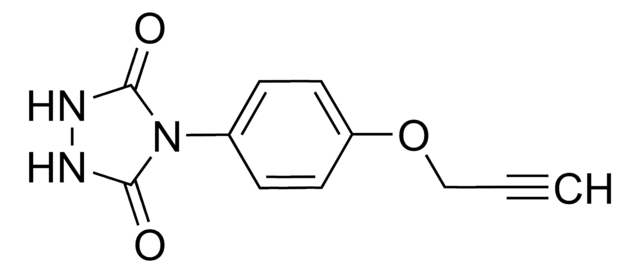


![Tris[(1-Benzyl-1H-1,2,3-Triazol-4-yl)methyl]amin 97%](/deepweb/assets/sigmaaldrich/product/structures/179/695/86a721c8-2a4c-4e4f-bc36-6276ce7a941f/640/86a721c8-2a4c-4e4f-bc36-6276ce7a941f.png)