Alle Fotos(2)
Wichtige Dokumente
539406
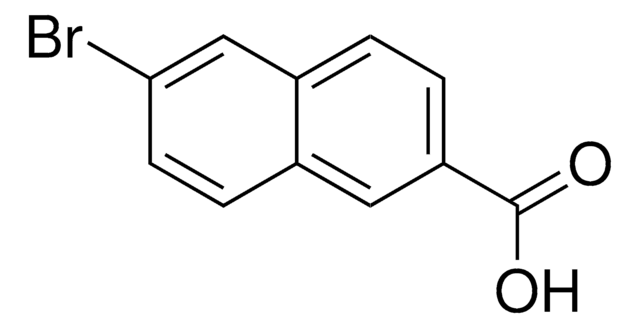
Methyl-6-brom-2-naphthoat
98%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Lineare Formel:
BrC10H6CO2CH3
CAS-Nummer:
Molekulargewicht:
265.10
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
98%
mp (Schmelzpunkt)
123-126 °C (lit.)
Funktionelle Gruppe
bromo
ester
SMILES String
COC(=O)c1ccc2cc(Br)ccc2c1
InChI
1S/C12H9BrO2/c1-15-12(14)10-3-2-9-7-11(13)5-4-8(9)6-10/h2-7H,1H3
InChIKey
JEUBRLPXJZOGPX-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
Methyl 6-bromo-2-naphthoate undergoes aromatic Finkelstein reaction followed by hydrolysis to afford 6-iodo-2-naphthoic acid.
Anwendung
Methyl 6-bromo-2-naphthoate may be used to synthesize:
- 6-vinyl-2-naphthalencarbaldehyde
- methyl 6-(3-tert-butyl-4-methoxyphenyl)-2-naphthoate
- methyl 6-[3-tert-butyl-4-[(tert-butyldiethylsilyl)oxy]-phenyl]-2-naphthoate
- methyl 6-[3-(1-adamantyl)-4-[(tert-butyldimethylsilyl)-oxy]phenyl]-2-naphthoate
- methyl 6-[3-(1-adamantyl)-4-[[(2,3-dimethyl-1,3-dioxolan-4-yl)methylloxy]phenyl]-2-naphthoate
- 2-bromo-6-(bromomethyl)naphthalene
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Carboxy-1, 4-phenylenevinylene-and carboxy-2, 6-naphthylene-vinylene unsymmetrical substituted zinc phthalocyanines for dye-sensitized solar cells.
Silvestri F, et al.
Journal of Porphyrins and Phthalocyanines, 13(03), 369-375 (2009)
B Charpentier et al.
Journal of medicinal chemistry, 38(26), 4993-5006 (1995-12-22)
The retinoic acid receptors (RARs) transduce retinoid dependant gene regulation, and many biological effects of retinoids are mediated through binding and activation of three closely related receptor subtypes (RAR alpha, RAR beta, and RAR gamma). In order to investigate the
Phil M Pithan et al.
Beilstein journal of organic chemistry, 12, 854-862 (2016-06-25)
Cationic biaryl derivatives were synthesized by Suzuki-Miyaura coupling of 3-bromonaphtho[1,2-b]quinolizinium bromide with arylboronic acids. The resulting cationic biaryl derivatives exhibit pronounced fluorosolvatochromic properties. First photophysical studies in different solvents showed that the emission energy of the biaryl derivatives decreases with
Mark W Irvine et al.
Journal of medicinal chemistry, 55(1), 327-341 (2011-11-25)
Competitive N-methyl-d-aspartate receptor (NMDAR) antagonists bind to the GluN2 subunit, of which there are four types (GluN2A-D). We report that some N(1)-substituted derivatives of cis-piperazine-2,3-dicarboxylic acid display improved relative affinity for GluN2C and GluN2D versus GluN2A and GluN2B. These derivatives
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.



![[1,1′-Bis(diphenylphosphino)ferrocen]dichlorpalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)



