Alle Fotos(3)
Wichtige Dokumente
433098
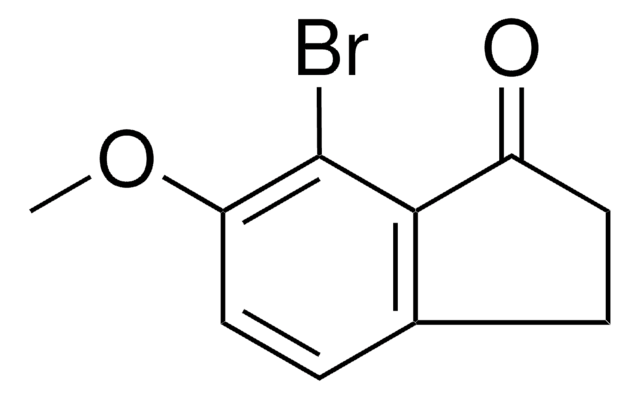
5-Brom-1-indanon
97%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(3)
About This Item
Empirische Formel (Hill-System):
C9H7BrO
CAS-Nummer:
Molekulargewicht:
211.06
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
97%
mp (Schmelzpunkt)
126-129 °C (lit.)
Funktionelle Gruppe
bromo
ketone
SMILES String
Brc1ccc2C(=O)CCc2c1
InChI
1S/C9H7BrO/c10-7-2-3-8-6(5-7)1-4-9(8)11/h2-3,5H,1,4H2
InChIKey
KSONICAHAPRCMV-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
5-Bromo-1-indanone is a 1-indanone derivative. Its physical properties like density, freezing point and refractive index have been determined. It participates in the synthesis of the imidazolyl and triazolyl substituted biphenyl compounds.
Signalwort
Warning
Gefahreneinstufungen
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Turn-On Fluorogenic Probes for the Selective and Quantitative Detection of the Cyanide Anion from Natural Sources.
Gomez T, et al.
Chemistry (Weinheim An Der Bergstrasse, Germany), 8(6), 1271-1278 (2013)
Yaws CL.
The Yaws Handbook of Physical Properties for Hydrocarbons and Chemicals, 229-229 (2015)
Andrew K Takle et al.
Bioorganic & medicinal chemistry letters, 16(2), 378-381 (2005-11-02)
A novel triarylimidazole derivative, SB-590885 (33), bearing a 2,3-dihydro-1H-inden-1-one oxime substituent has been identified as a potent and extremely selective inhibitor of B-Raf kinase.
Y Zhuang et al.
Bioorganic & medicinal chemistry, 8(6), 1245-1252 (2000-07-15)
The synthesis of a new series of P450 17 inhibitors is described. The imidazol-1-yl compounds 5 showed strong inhibition of P450 17 rat and especially human enzyme, the most active compounds being 5ax, 5ay and 5bx with IC50 values of
Agustin Casimiro-Garcia et al.
Journal of medicinal chemistry, 54(12), 4219-4233 (2011-05-12)
Mining of an in-house collection of angiotensin II type 1 receptor antagonists to identify compounds with activity at the peroxisome proliferator-activated receptor-γ (PPARγ) revealed a new series of imidazo[4,5-b]pyridines 2 possessing activity at these two receptors. Early availability of the
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.