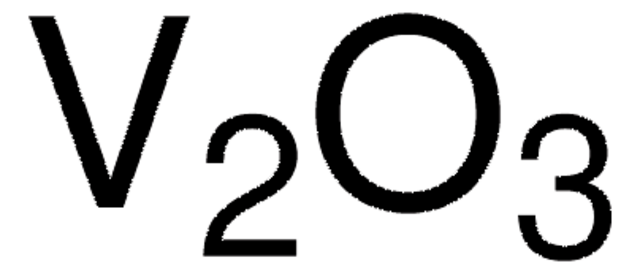Alle Fotos(3)
Wichtige Dokumente
215066
Gallium(III)-oxid
≥99.99% trace metals basis
Synonym(e):
Gallium trioxide
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(3)
About This Item
Empirische Formel (Hill-System):
Ga2O3
CAS-Nummer:
Molekulargewicht:
187.44
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352303
PubChem Substanz-ID:
NACRES:
NA.23
Empfohlene Produkte
Qualitätsniveau
Assay
≥99.99% trace metals basis
Form
(crystalline powder)
Eignung der Reaktion
reagent type: catalyst
core: gallium
Dichte
5.88 g/mL at 25 °C
SMILES String
O=[Ga]O[Ga]=O
InChI
1S/2Ga.3O
InChIKey
QZQVBEXLDFYHSR-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Gallium(III) oxide (Ga2O3) is a wide band gap semiconductor that belongs to a family of transparent semiconducting oxides (TSO). It can form different polymorphs such as α-,β-, γ-, δ-, and ε-. Polycrystalline and nanocrystalline Ga2O3 can be prepared using several methods such as chemical vapor deposition, thermal vaporization, and sublimation, molecular beam epitaxy, melt growth, etc. It is widely used as a functional material in various applications including optoelectronics, chemical sensors, catalysis, semiconductor devices, field-effect transistors, and many others.
Anwendung
Starting material for the preparation of Sr2CuGaO3S, an example of a rare square pyramidal gallium.
Ga2O3 is widely used as a host material for the fabrication of electroluminescent devices. For example, europium-doped Ga2O3 thin films can be used as a light-emitting layer to fabricate an optically transparent electroluminescent device.
Due to its distinct optical and electrical properties like moderate conductivity and high laser damage threshold, Ga2O3 can be used in laser-driven electron accelerators, low-loss plasmonics, and Si-based dielectric laser accelerators.
It can also be used as an effective catalyst for the dehydrogenation of propane to propene.
Due to its distinct optical and electrical properties like moderate conductivity and high laser damage threshold, Ga2O3 can be used in laser-driven electron accelerators, low-loss plasmonics, and Si-based dielectric laser accelerators.
It can also be used as an effective catalyst for the dehydrogenation of propane to propene.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 2
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
G Wilma Busser et al.
ChemSusChem, 5(11), 2200-2206 (2012-10-24)
The deposition of hydrogen evolution sites on photocatalysts is a crucial step in the multistep process of synthesizing a catalyst that is active for overall photocatalytic water splitting. An alternative approach to conventional photodeposition was developed, applying the photocatalytic reforming
The rate of charge tunneling through self-assembled monolayers is insensitive to many functional group substitutions.
Hyo Jae Yoon et al.
Angewandte Chemie (International ed. in English), 51(19), 4658-4661 (2012-04-17)
Farheen N Sayed et al.
Journal of nanoscience and nanotechnology, 11(4), 3363-3369 (2011-07-23)
We report on the structural and magnetic properties of nanoparticles of NiGa2O4 and 5 at.% M doped (M = Mn2+, Cu2+, Co2+, Fe3+ and Tb3+) at Ga site of NiGa2O4, synthesized by gel-combustion method. The particle size, as investigated by
Naoya Kumagai et al.
Chemical communications (Cambridge, England), 47(6), 1884-1886 (2010-12-07)
Using the Rh(3+) ion (Rh d(6)) in a regular octahedral coordination, which forms fully occupied t(2g)(6) and empty e(g)(0) as a result of ligand-field splitting, we demonstrated that Rh-doped ZnGa(2)O(4) had midgap states created by t(2g)(6) and e(g)(0) that had
Sr(2)CuGaO(3)S, a Rare Example of Square Pyramidal Gallium.
W. J. Zhu et al.
Inorganic chemistry, 36(17), 3576-3577 (1997-08-13)
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.