Fortfahren mit
Anmelden zur Ansicht der Organisations- und Vertragspreise.
Größe auswählen
Ansicht ändern
| Ihnen/SKU | Verfügbarkeit | Preis |
|---|
Über diesen Artikel
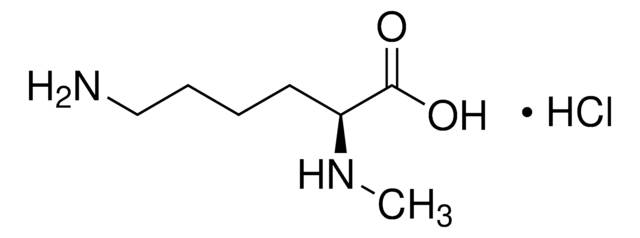
Empirische Formel (Hill-System):
C6H11NO2
CAS-Nummer:
Molekulargewicht:
129.16
UNSPSC Code:
12352209
NACRES:
NA.22
PubChem Substance ID:
MDL number:
Beilstein/REAXYS Number:
4350211
Preise und Verfügbarkeit sind derzeit nicht verfügbar.
Technischer Dienst
Benötigen Sie Hilfe? Unser Team von erfahrenen Wissenschaftlern ist für Sie da.
Unterstützung erhaltenQuality Segment
assay
≥98.0% (TLC)
reaction suitability
reaction type: solution phase peptide synthesis
application(s)
peptide synthesis
SMILES string
C[C@]1(CCCN1)C(O)=O
InChI
1S/C6H11NO2/c1-6(5(8)9)3-2-4-7-6/h7H,2-4H2,1H3,(H,8,9)/t6-/m0/s1
InChI key
LWHHAVWYGIBIEU-LURJTMIESA-N
1 of 1
Dieser Artikel | |||
|---|---|---|---|
| assay ≥98.0% (TLC) | assay ≥98.0% (TLC) | assay ≥97.0% (TLC) | assay ≥98.0% (TLC) |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| reaction suitability reaction type: solution phase peptide synthesis | reaction suitability - | reaction suitability reaction type: solution phase peptide synthesis | reaction suitability reaction type: solution phase peptide synthesis |
| application(s) peptide synthesis | application(s) - | application(s) peptide synthesis | application(s) peptide synthesis |
Lagerklasse
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.