All Photos(1)
About This Item
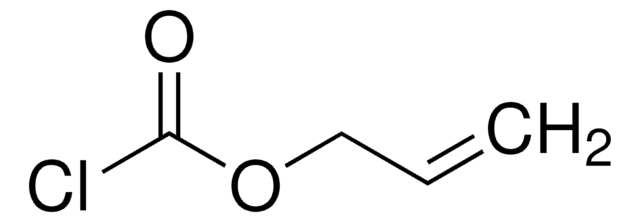
Empirical Formula (Hill Notation):
C8H9NO5
CAS Number:
Molecular Weight:
199.16
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
96%
form
liquid
refractive index
n20/D 1.482
density
1.287 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
C=CCOC(=O)ON1C(=O)CCC1=O
InChI
1S/C8H9NO5/c1-2-5-13-8(12)14-9-6(10)3-4-7(9)11/h2H,1,3-5H2
InChI key
OIXALTPBNZNFLJ-UHFFFAOYSA-N
Related Categories
Application
N-(Allyloxycarbonyloxy)succinimide (alloc-Su) can be used as:
- A building block for the preparation of glycopeptide scaffolds.
- A reagent in the synthesis of various functional cyclic carbonate monomers from 2-amino-1,3-propane diols.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
242.6 °F
Flash Point(C)
117 °C
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Synthesis and in vitro evaluation of bisphosphonated glycopeptide prodrugs for the treatment of osteomyelitis
Tanaka KSE, et al.
Bioorganic & medicinal chemistry letters, 20(4), 1355-1359 (2010)
2-Amino-1, 3-propane diols: a versatile platform for the synthesis of aliphatic cyclic carbonate monomers
Venkataraman S, et al.
Polym. Chem., 4(10), 2945-2948 (2013)
Bo Wu et al.
Molecules (Basel, Switzerland), 25(19) (2020-10-07)
Four bis-lactam [i, i+4]-stapled peptides with d- or l-α-methyl-thialysines were constructed on a model peptide sequence derived from p110α[E545K] and subjected to circular dichroism (CD) and proteolytic stability assessment, alongside the corresponding bis-lactam [i, i+4]-stapled peptide with l-thialysine. The %
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service