All Photos(1)
About This Item
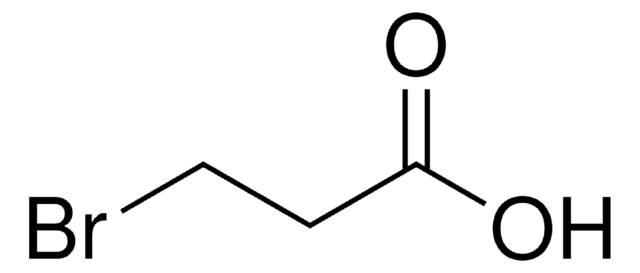
Linear Formula:
H2NC4H6CO2H
CAS Number:
Molecular Weight:
115.13
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
reaction suitability
reaction type: solution phase peptide synthesis
mp
261 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
NC1(CCC1)C(O)=O
InChI
1S/C5H9NO2/c6-5(4(7)8)2-1-3-5/h1-3,6H2,(H,7,8)
InChI key
FVTVMQPGKVHSEY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Insulin-induced phospho-oligosaccharide stimulates amino acid transport in isolated rat hepatocytes.
I Varela et al.
The Biochemical journal, 267(2), 541-544 (1990-04-15)
The ability of the insulin-induced phospho-oligosaccharide to stimulate amino acid transport was studied in isolated rat hepatocytes. At low alpha-aminoisobutyric acid concentrations (0.1 mM), both 100 nM-insulin and 10 microM-phospho-oligosaccharide doubled amino acid uptake after 2 h of incubation. This
1-Aminocyclobutane-1-carboxylate (ACBC): a specific antagonist of the N-methyl-D-aspartate receptor coupled glycine receptor.
W F Hood et al.
European journal of pharmacology, 161(2-3), 281-282 (1989-02-28)
T S Rao et al.
Neuropharmacology, 29(3), 305-309 (1990-03-01)
Following intravenous administration, 1-aminocyclobutane-1-carboxylate (ACBC, 100 mg/kg), a N-methyl-D-aspartate (NMDA)-associated glycine receptor antagonist, was eliminated with a T1/2 of 5 min in mouse brain and 4 min in rat cerebrospinal fluid (CSF). 1-Aminocyclopropane-1-carboxylate (ACC), a NMDA-associated glycine receptor agonist, was
R P Compton et al.
European journal of pharmacology, 188(1), 63-70 (1990-01-23)
Activation of the N-methyl-D-aspartate (NMDA) receptor complex is subject to modulation via interactions at a coupled [3H]glycine recognition site in rat brain synaptic plasma membranes (SPM). We examined the effect of the potent and specific glycine site antagonists, 1-hydroxy-3-amino-2-pyrrolidone (HA-966)
V N Balaji et al.
Peptide research, 8(3), 178-186 (1995-05-01)
Conformationally constrained peptidomimetics are being increasingly used in the development of 3-D pharmacophores of peptide-based drug candidates and to alter their metabolic stability towards achievements of oral bioavailability. Here we present conformational energy calculations on model compounds containing 1-aminocyclobutane carboxylic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service