1A01200
USP
Rosuvastatin Impurity D
Pharmaceutical Analytical Impurity (PAI)
동의어(들):
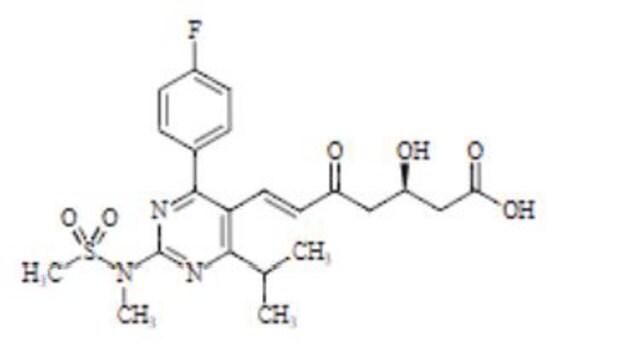
N-[4-(4-fluorophenyl)-5-[(E)-2-[(2S,4R)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl]ethenyl]-6-(1-methylethyl)pyrimidin-2-yl]-N-methylmethanesulfonamide
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C22H26FN3O5S
CAS Number:
Molecular Weight:
463.52
UNSPSC 코드:
41116107
NACRES:
NA.24
추천 제품
Grade
pharmaceutical analytical impurity (PAI)
Agency
USP
API family
rosuvastatin
제조업체/상표
USP
응용 분야
pharmaceutical
형식
neat
저장 온도
2-8°C
InChI
1S/C22H26FN3O5S/c1-13(2)20-18(10-9-17-11-16(27)12-19(28)31-17)21(14-5-7-15(23)8-6-14)25-22(24-20)26(3)32(4,29)30/h5-10,13,16-17,27H,11-12H2,1-4H3/b10-9+/t16-,17-/m1/s1
InChI key
SOEGVMSNJOCVHT-VEUZHWNKSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Rosuvastatin Impurity D is a USP Pharmaceutical Analytical Impurity (PAI).
USP PAI are a product line of impurities suitable for research and analytical purposes, which help to ensure the quality and safety of medicines.
Associated Drug Substance: Rosuvastatin Calcium
Therapeutic Area: Antihyperlipidemics
For more information about this PAI, visit here.
USP PAI are a product line of impurities suitable for research and analytical purposes, which help to ensure the quality and safety of medicines.
Associated Drug Substance: Rosuvastatin Calcium
Therapeutic Area: Antihyperlipidemics
For more information about this PAI, visit here.
애플리케이션
Rosuvastatin Impurity D (USP PAI) is intended for use in analytical testing to detect, identify, and measure pharmaceutical impurities.
특징 및 장점
USP PAI advance your early analytical R&D and process development. PAI can be used in the following applications:
1. Conduct analytical tests during early formulation feasibility studies.
2. Determine degradation impurities produced during stress studies.
3. Develop, validate, and transfer analytical methods.
4. Perform spiking studies during process R&D to demonstrate depletion upon recrystallization.
5. Record retention times and/or spectra
6. Determine relative response factors.
7. Identify unknown impurities that formed during ICH stability conditions.
8. Identify impurities that are present in the Reference Listed Drug
9. Test for and profile impurities not listed in drug substance and drug product monographs.
1. Conduct analytical tests during early formulation feasibility studies.
2. Determine degradation impurities produced during stress studies.
3. Develop, validate, and transfer analytical methods.
4. Perform spiking studies during process R&D to demonstrate depletion upon recrystallization.
5. Record retention times and/or spectra
6. Determine relative response factors.
7. Identify unknown impurities that formed during ICH stability conditions.
8. Identify impurities that are present in the Reference Listed Drug
9. Test for and profile impurities not listed in drug substance and drug product monographs.
분석 메모
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
기타 정보
Sales restrictions may apply.
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
STOT RE 2 Oral
표적 기관
Liver,Kidney
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.