PHR1658
Dimenhydrinate
Pharmaceutical Secondary Standard; Certified Reference Material
동의어(들):
Dimenhydrinate, DMH, N-(2-Diphenylmethoxyethyl)-N,N-dimethylammonium 8-chlorotheophyllinate
About This Item
추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to Ph. Eur. D2000000
traceable to USP 1206006
API family
dimenhydrinate
CofA
current certificate can be downloaded
포장
pkg of 1 g
기술
HPLC: suitable
gas chromatography (GC): suitable
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-30°C
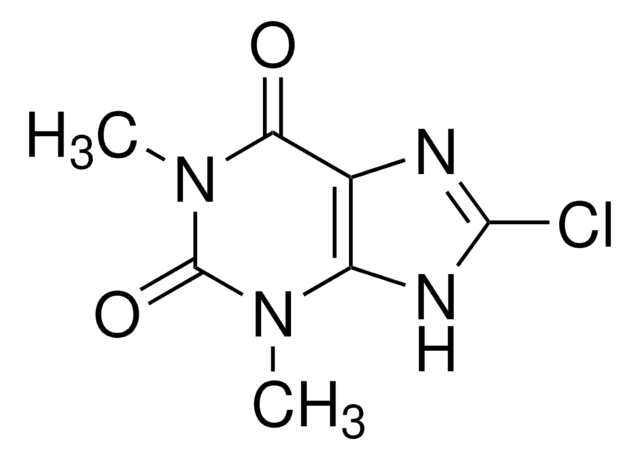
SMILES string
CN1C(=O)N(C)c2nc(Cl)[nH]c2C1=O.CN(C)CCOC(c3ccccc3)c4ccccc4
InChI
1S/C17H21NO.C7H7ClN4O2/c1-18(2)13-14-19-17(15-9-5-3-6-10-15)16-11-7-4-8-12-16;1-11-4-3(9-6(8)10-4)5(13)12(2)7(11)14/h3-12,17H,13-14H2,1-2H3;1-2H3,(H,9,10)
InChI key
NFLLKCVHYJRNRH-UHFFFAOYSA-N
유전자 정보
human ... HRH1(3269)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Dimenhydrinate, a diphenhydramine salt of 8-chlorotheophylline, exhibits antihistaminic activity, with antimuscarinic and sedative effects. It finds its major application as an antiemetic drug in the prevention and treatment of motion sickness.
애플리케이션
분석 메모
기타 정보
각주
추천 제품
관련 제품
Choose from one of the most recent versions:
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.