8.51006
HBTU
≥99.0% (HPLC), for peptide synthesis, Novabiochem®
동의어(들):
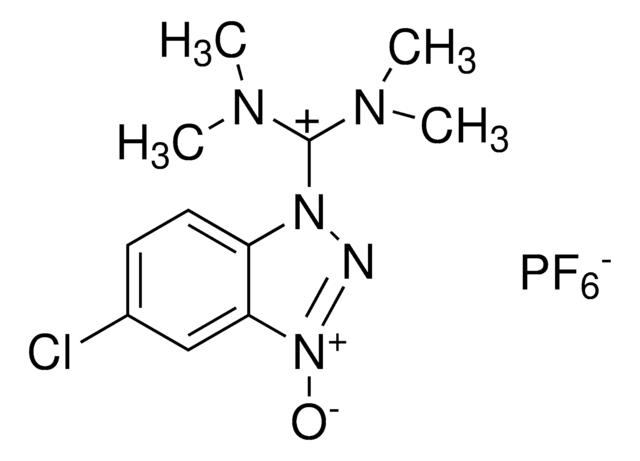
HBTU, 2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethylaminium hexafluorophosphate
About This Item
추천 제품
product name
HBTU, 2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate Novabiochem®
Quality Level
제품 라인
Novabiochem®
분석
≥99.0% (HPLC)
형태
powder
효능
>2000 mg/kg LD50, oral (Rat)
반응 적합성
reaction type: Coupling Reactions
제조업체/상표
Novabiochem®
pH
4.1 (1.6 g/L in H2O)
mp
250 °C
solubility
1.6 g/L
응용 분야
peptide synthesis
저장 온도
2-8°C
InChI
1S/C11H16N5O/c1-14(2)11(15(3)4)17-16-10-8-6-5-7-9(10)12-13-16/h5-8H,1-4H3/q+1
InChI key
CLZISMQKJZCZDN-UHFFFAOYSA-N
일반 설명
Associated Protocols and Technical Articles
Guide to Selection of Coupling Reagents
Literature references
[1] R. Knorr, et al. (1989) Tetrahedron Lett., 30, 1927.
[2] M. S. Bernatowicz, et al. (1989) Tetrahedron Lett., 30, 4645.
[3] D. Ambrosius, et al. (1989) Biol. Chem. Hoppe-Seyler, 370, 217.
[4] C. G. Fields, et al. (1991) Pept. Res., 4, 95.
[5] A. G. Beck-Sickinger, et al. (1991) Pept. Res., 4, 88.
[6] G. E. Reid, et al. (1992) Anal. Biochem., 200, 301.
[7] G. B. Fields, et al. in ′Innovation & Perspectives in Solid Phase Synthesis, 1st International Symposium′, R. Epton (Eds), SPCC UK Ltd., Birmingham, 1990, pp. 241.
[8] P. A. Baybayan, et al. in ′Peptides, Chemistry & Biology, Proc. 12th American Peptide Symposium′, J. A. Smith & J. E. Rivier (Eds), ESCOM, Leiden, 1992, pp. 566.
[9] J. J. Dudash, et al. (1993) Synth. Commun., 23, 349.
애플리케이션
- Synthesis of Quinoxaline Derivatives Using HBTU: A study highlighting the use of HBTU as a Lewis acid catalyst for synthesizing quinoxaline derivatives, presenting a mild and green protocol (Popatkar and Meshram, 2020).
- Efficient Conversion of Carboxylic Acids into Benzimidazoles: Details an HBTU-promoted methodology for converting carboxylic acids into benzimidazoles in a one-pot strategy (Barasa and Yoganathan, 2018).
- Synthesis of Malonyl-linked Glycoconjugates: Discusses the use of HBTU in the synthesis of glycoconjugates, comparing its efficiency with other reagents (Nörrlinger et al., 2016).
결합
분석 메모
Appearance of substance (visual): powder
Identity (IR): passes test
Assay (HPLC, area%): ≥ 99.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
법적 정보
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Skin Sens. 1A
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
문서
Novabiochem® coupling reagents for in situ activation are fast-reacting and compatible with various amino acids.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.









![COMU 1-[(1-(Cyano-2-ethoxy-2-oxoethylideneaminooxy) dimethylaminomorpholino)] uronium hexafluorophosphate Novabiochem®](/deepweb/assets/sigmaaldrich/product/images/237/337/13566c06-8931-4cc2-8621-c8742a392cd6/640/13566c06-8931-4cc2-8621-c8742a392cd6.jpg)