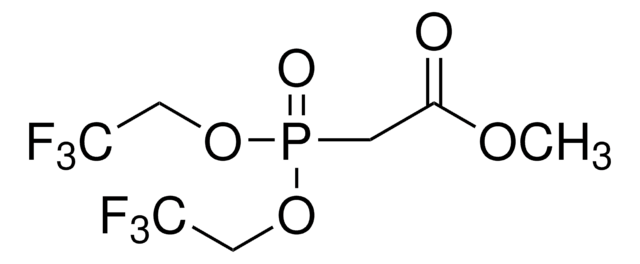
405957
Tris(1,1,1,3,3,3-hexafluoro-2-propyl) phosphite
98%
동의어(들):
Tris(2H-perfluoro-2-propyl) phosphite
크기 선택
About This Item
추천 제품
Quality Level
분석
98%
양식
liquid
refractive index
n20/D 1.300 (lit.)
bp
130 °C (lit.)
density
1.69 g/mL at 25 °C (lit.)
작용기
fluoro
SMILES string
FC(F)(F)C(OP(OC(C(F)(F)F)C(F)(F)F)OC(C(F)(F)F)C(F)(F)F)C(F)(F)F
InChI
1S/C9H3F18O3P/c10-4(11,12)1(5(13,14)15)28-31(29-2(6(16,17)18)7(19,20)21)30-3(8(22,23)24)9(25,26)27/h1-3H
InChI key
MJOVEPJSFHDSOJ-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
일반 설명
애플리케이션
- Preparation of new phosphonylating and coupling agents for the synthesis of oligodeoxyribonucleotides.[2]
- Preparation of nucleoside H-phosphonate units.[4]
- Synthesis of tetrakis(1,1,1,3,3,3-hexafluoroisopropoxy)(phenylthio)phosphorane by reacting with 1,1,1,3,3,3-hexafluoroisopropyl benzenesulfenate.[5]
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
활성 필터
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.



![4-tert-Butylcalix[4]arene 95%](/deepweb/assets/sigmaaldrich/product/structures/141/993/6110bb7e-274f-45f8-887a-f1841a7dae7c/640/6110bb7e-274f-45f8-887a-f1841a7dae7c.png)