추천 제품
분석
98%
형태
solid
bp
178-180 °C/60 mmHg (lit.)
mp
65-67 °C (lit.)
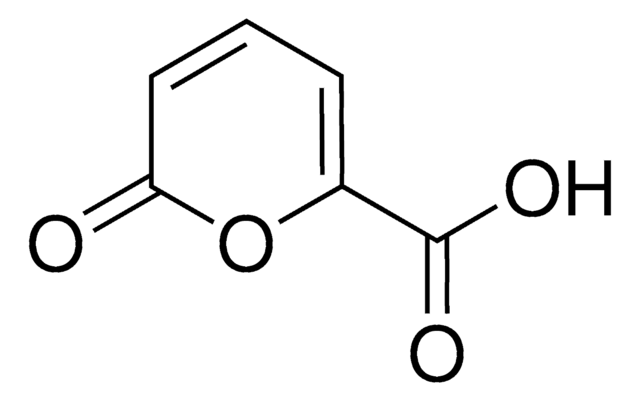
SMILES string
COC(=O)C1=COC(=O)C=C1
InChI
1S/C7H6O4/c1-10-7(9)5-2-3-6(8)11-4-5/h2-4H,1H3
InChI key
HHWWWZQYHPFCBY-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
일반 설명
Methyl coumalate is a 2-pyrone and acts as dienophile in Diels-Alder reaction. It reacts with 1,3-butadienes at 100°C to yield tetrahydrocoumarins and 4-methoxycarbonyltricyclo[3.2.1.02,7]octenes. It also reacts with cyclohexadiene to afford tetrahydronaphthalene-2-carboxylate. It undergoes Diels-Alder reaction with unactivated alkenes to afford para-substituted adducts.
애플리케이션
Methyl coumalate has been used as reagent in phosphine-catalyzed [4+3] annulation of modified allylic carbonates. It was also used in the preparation of 7-carboxyquinolizinium derivatives.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
Aromatics from pyrones: para-substituted alkyl benzoates from alkenes, coumalic acid and methyl coumalate.
Kraus GA, et al.
Green Chemistry, 13(10), 2734-2736 (2011)
Quinolizinium Compounds by Cyclization of Pyridones from Methyl Coumalate and ?-Phenylethylamines.
Wiley RH, et al.
Journal of the American Chemical Society, 75(18), 4482-4484 (1953)
Diels-Alder reaction of methyl coumalate with 1, 3-dienes.
Imagawa T, et al.
Tetrahedron, 30(14), 2227-2231 (1974)
Suqing Zheng et al.
Organic letters, 11(17), 3978-3981 (2009-08-13)
A phosphine-catalyzed [4 + 3] annulation of modified allylic carbonates with methyl coumalate was developed. This strategy offered a powerful method for the construction of bicyclo[3.2.2]nonadiene skeleton with high stereoselectivity.
문서
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.