おすすめの製品
product name
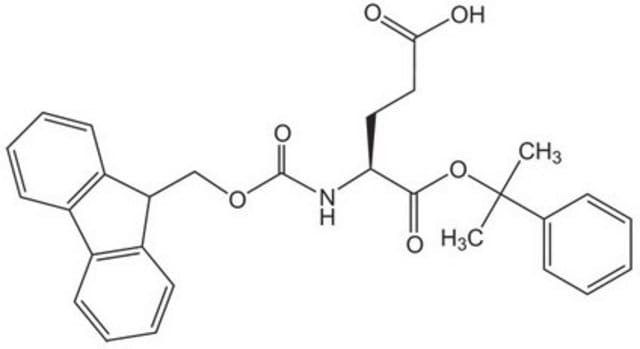
Fmoc-Glu(O-2-PhiPr)-OH, Novabiochem®
品質水準
製品種目
Novabiochem®
アッセイ
≥90.0% (acidimetric)
≥95.0% (HPLC)
≥97% (TLC)
形状
powder
反応適合性
reaction type: Fmoc solid-phase peptide synthesis
メーカー/製品名
Novabiochem®
アプリケーション
peptide synthesis
官能基
carboxylic acid
保管温度
15-25°C
InChI
1S/C29H29NO6/c1-29(2,19-10-4-3-5-11-19)36-26(31)17-16-25(27(32)33)30-28(34)35-18-24-22-14-8-6-12-20(22)21-13-7-9-15-23(21)24/h3-15,24-25H,16-18H2,1-2H3,(H,30,34)(H,32,33)/t25-/m0/s1
InChI Key
NPNRKQDJIWMHNG-VWLOTQADSA-N
関連するカテゴリー
詳細
Quasi-orthogonally-protected Glu derivative. The 2-PhiPr group can be removed selectively in the presence of tBu-based protecting groups by treatment with 1% TFA in DCM [1], making this derivative an extremely useful tool for the preparation of cyclic peptides by Fmoc SPPS [2] or for library synthesis. For the on-resin synthesis of side-chain to side-chain lactam bridged peptides, the combination of Lys(Mtt) and Glu(O-2-PhiPr) is particularly advantageous since both side-chains can be simultaneously unmasked in a single step.
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Literature references
[1] C. Yue, et al. (1993) Tetrahedron Lett., 34, 323.
[2] F. Dick, et al. in ′Peptides 1996, Proc. of 24th European Peptide Symposium′, R. Ramage & R. Epton (Eds), Mayflower Scientific Ltd., Birmingham, 1998, pp. 339.
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Literature references
[1] C. Yue, et al. (1993) Tetrahedron Lett., 34, 323.
[2] F. Dick, et al. in ′Peptides 1996, Proc. of 24th European Peptide Symposium′, R. Ramage & R. Epton (Eds), Mayflower Scientific Ltd., Birmingham, 1998, pp. 339.
関連事項
Replaces: 04-12-1199
アナリシスノート
Color (visual): white to slight yellow to beige
Appearance of substance (visual): powder
Identity (IR): passes test
Enantiomeric purity: ≥ 99.0 % (a/a)
Purity (TLC(011A)): ≥ 97 %
Purity (TLC(0811)): ≥ 97 %
Assay (HPLC, area%): ≥ 95.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Assay (acidimetric): ≥ 90.0 %
Water (K. F.): ≤ 2.0 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
Appearance of substance (visual): powder
Identity (IR): passes test
Enantiomeric purity: ≥ 99.0 % (a/a)
Purity (TLC(011A)): ≥ 97 %
Purity (TLC(0811)): ≥ 97 %
Assay (HPLC, area%): ≥ 95.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Assay (acidimetric): ≥ 90.0 %
Water (K. F.): ≤ 2.0 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
法的情報
Novabiochem is a registered trademark of Merck KGaA, Darmstadt, Germany
保管分類コード
11 - Combustible Solids
WGK
WGK 2
引火点(°F)
Not applicable
引火点(℃)
Not applicable
試験成績書(COA)
製品のロット番号・バッチ番号を入力して、試験成績書(COA) を検索できます。ロット番号・バッチ番号は、製品ラベルに「Lot」または「Batch」に続いて記載されています。
資料
Novabiochem® offers orthogonally protected amino acids for peptide synthesis, including cyclic and branched peptides.
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)