730998
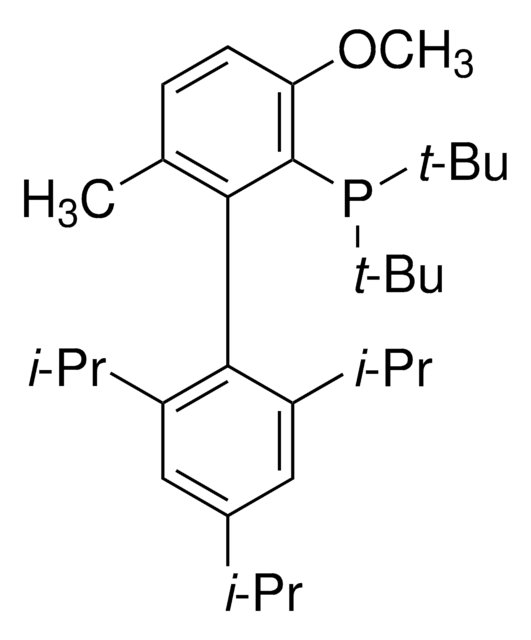
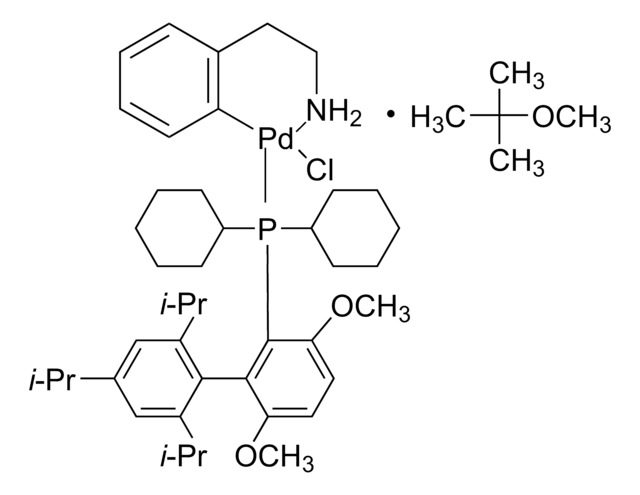
tBuBrettPhos
97%
Synonym(s):
t-Bu Brett Phos, t-BuBrett-Phos, tertButylBrettPhos, 2-(Di-tert-butylphosphino)-2′,4′,6′- triisopropyl-3,6-dimethoxy-1,1′-biphenyl, t-BuBrett Phos, t-BuBrettPhos, [3,6-Dimethoxy-2′,4′,6′-tris(1-methylethyl) [1,1′-biphenyl]-2-yl]bis(1,1-dimethylethyl)phosphine, tert-ButylBrettPhos
About This Item
Recommended Products
Quality Level
Assay
97%
form
solid
reaction suitability
reaction type: Cross Couplings
reagent type: ligand
reaction type: Arylations
reagent type: ligand
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reagent type: ligand
reaction type: C-X Bond Formation
reagent type: ligand
reaction type: Fluorinations
mp
166-170 °C
functional group
phosphine
SMILES string
COc1ccc(OC)c(c1P(C(C)(C)C)C(C)(C)C)-c2c(cc(cc2C(C)C)C(C)C)C(C)C
InChI
1S/C31H49O2P/c1-19(2)22-17-23(20(3)4)27(24(18-22)21(5)6)28-25(32-13)15-16-26(33-14)29(28)34(30(7,8)9)31(10,11)12/h15-21H,1-14H3
InChI key
REWLCYPYZCHYSS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
tBuBrettPhos is a phosphine ligand widely used in palladium-catalyzed cross-coupling reactions.
Application
- Buchwald-Hartwig amination and C-O coupling
- Suzuki, Negishi, Stille, Hiyama, Sonogashira cross-couplings
- α-Arylation reaction
New Applications:
- Conversion of aryl and vinyl triflates to bromides and chlorides
- Conversion of aryl triflates to aryl fluorides
- O-Arylation of ethyl acetohydroximate
- Conversion of aryl chlorides and sulfonates to nitroaromatics
Features and Benefits
- White crystalline solid
- Air- and moisture-stable
- Thermally stable
- Highly efficient
- Wide functional group tolerance
- Excellent selectivity and conversion
Legal Information
related product
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
ISHL Indicated Name
Substances Subject to be Indicated Names
ISHL Notified Names
Substances Subject to be Notified Names
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
The Catalexis platform enhances catalysis by digitally optimizing catalyst selection to identify the most effective phosphine ligands for cross-coupling reactions.
Explore reliable, premium grade catalysis materials for your pharma or industrial project. Specialty chemicals and formulations are available in bulk quantities and volumes from a few grams to multi-metric tons with complete documentation to simplify your leap from development to commercialization.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service








![5-(Di-tert-butylphosphino)-1′, 3′, 5′-triphenyl-1′H-[1,4′]bipyrazole 97%](/deepweb/assets/sigmaaldrich/product/structures/137/599/8b2f4b58-3384-40aa-9295-0887f7985525/640/8b2f4b58-3384-40aa-9295-0887f7985525.png)