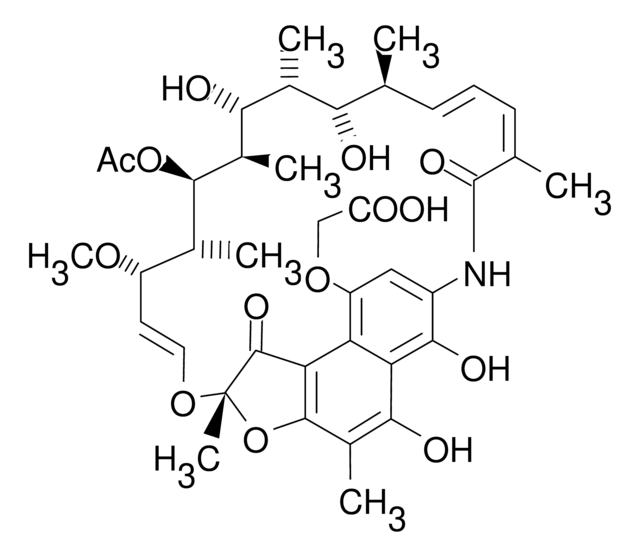
R0950000
Rifamycin S
European Pharmacopoeia (EP) Reference Standard
Sinonimo/i:
1,4-Dideoxy-1,4-dihydro-1,4-dioxorifamycin
About This Item
Prodotti consigliati
Grado
pharmaceutical primary standard
Famiglia di API
rifamycin, rifampicin
Produttore/marchio commerciale
EDQM
applicazioni
pharmaceutical (small molecule)
Formato
neat
Temperatura di conservazione
2-8°C
InChI
1S/C37H45NO12/c1-16-11-10-12-17(2)36(46)38-23-15-24(40)26-27(32(23)44)31(43)21(6)34-28(26)35(45)37(8,50-34)48-14-13-25(47-9)18(3)33(49-22(7)39)20(5)30(42)19(4)29(16)41/h10-16,18-20,25,29-30,33,41-43H,1-9H3,(H,38,46)/b11-10+,14-13+,17-12-/t16-,18+,19+,20+,25-,29-,30+,33+,37-/m0/s1
BTVYFIMKUHNOBZ-ODRIEIDWSA-N
Descrizione generale
Applicazioni
Confezionamento
Altre note
Prodotti correlati
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Ci dispiace, ma al momento non ci sono COA disponibili online per questo prodotto.
Se ti serve aiuto, non esitare a contattarci Servizio Clienti
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.