74011
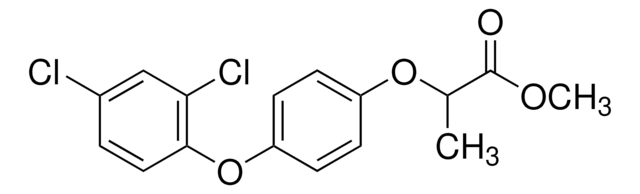
(±)-Diclofop
certified reference material, TraceCERT®, Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
Sinonimo/i:
(RS)-2-[4-(2,4-Dichlorophenoxy)phenoxy]propionic acid
About This Item
Prodotti consigliati
Grado
certified reference material
TraceCERT®
Livello qualitativo
Nome Commerciale
TraceCERT®
Durata
limited shelf life, expiry date on the label
Produttore/marchio commerciale
Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
Formato
neat
Temperatura di conservazione
2-8°C
Stringa SMILE
CC(Oc1ccc(Oc2ccc(Cl)cc2Cl)cc1)C(O)=O
InChI
1S/C15H12Cl2O4/c1-9(15(18)19)20-11-3-5-12(6-4-11)21-14-7-2-10(16)8-13(14)17/h2-9H,1H3,(H,18,19)
OOLBCHYXZDXLDS-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Certified content by quantitative NMR incl. uncertainty and expiry date are given on the certificate.
Download your certificate at: http://www.sigma-aldrich.com
(+/–)-Diclofop is a chiral herbicide that belongs to the chemical class of chiral aryloxyphenoxypropionate compounds. The actual herbicidal active ingredient― carboxylic acid, is released after application through hydrolysis of the ester. It is absorbed mainly through leaves and inhibits the biosynthesis of fatty acids by suppressing the activity of acetyl CoA carboxylase (ACCase). It is used for the post-emergence control of wild oats, wild millets, and other annual grass weeds commonly occurring in wheat, barley, rye, red fescue, and broad-leaved crops.
It was included on 1st June 2011 in Annex I of Directive 91/414/EEC by the European Commission Directive 2011/45/EU. It is authorized for use under EC Regulation No 1107/2009, as per the Commission Implementing Regulation (EU) No 540/2011, however it is a candidate for substitution.
Applicazioni
The (+/–)-Diclofop CRM can also be used as following:
- To evaluate the likely enantioselective oxidative stress produced in Microcystis aeruginosa by diclofop acid
- For analyzing the phytotoxic effects of diclofop acid enantiomers on the plant Arabidopsis thaliana
- To study the enantioselective toxicity of diclofop acid on the non-target rice Xiushui 63 seedlings
- In the chiral separation of diclofop-acid using one- and two- dimensional HPLC methods
- To determine the enantioselective toxicity and degradation of diclofop in three algal cultures
Prodotti consigliati
Note legali
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Choose from one of the most recent versions:
Certificati d'analisi (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.