8.02954
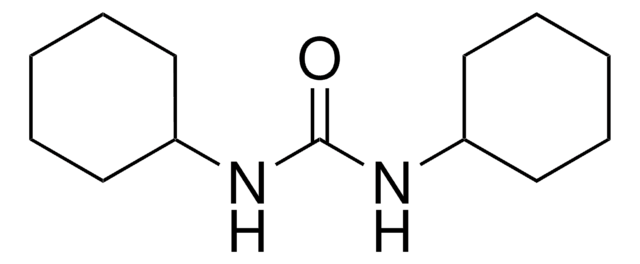
N,N′-Dicyclohexylcarbodiimide
for peptide synthesis
Sinónimos:
N,N′-Dicyclohexylcarbodiimide, DCC
About This Item
Productos recomendados
product name
N,N′-Dicyclohexylcarbodiimide, for synthesis
Quality Level
form
solid
potency
1110 mg/kg LD50, oral (Rat)
71 mg/kg LD50, skin (Rat)
reaction suitability
reaction type: Coupling Reactions
bp
148-152 °C/15 hPa
mp
35-36 °C
transition temp
flash point 113 °C
density
0.95 g/cm3 at 40 °C
bulk density
920 kg/m3
application(s)
peptide synthesis
storage temp.
2-30°C
InChI
1S/C13H22N2/c1-3-7-12(8-4-1)14-11-15-13-9-5-2-6-10-13/h12-13H,1-10H2
InChI key
QOSSAOTZNIDXMA-UHFFFAOYSA-N
Categorías relacionadas
General description
Application
- The synthesis of optically pure N-acyl-N,N′-dicyclohexylureas.
- The activation of the carboxylic acid groups in aromatic carboxylic acids to facilitates their reaction with (N-isocyanimino)trifluoroacetamide to form the corresponding 1,3,4-oxadiazole derivatives.
- The synthesis of poly (vinyl alcohol-co-vinyl levulinate) copolymers for use in biomedical applications.
signalword
Danger
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Skin Sens. 1
Storage Class
6.1D - Non-combustible, acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
wgk_germany
WGK 3
flash_point_f
235.4 °F - closed cup
flash_point_c
113 °C - closed cup
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico