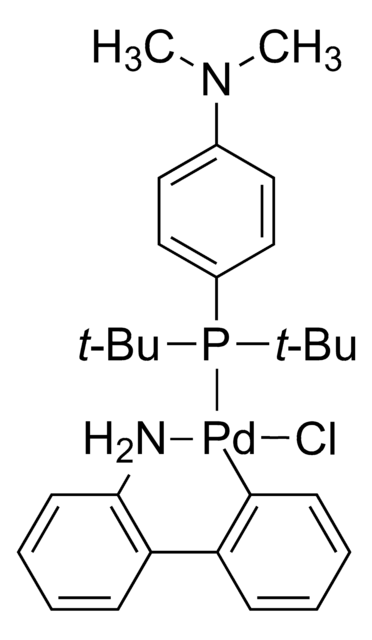
718750
BrettPhos Pd G1, Methyl t-Butyl Ether Adduct
may contain up to 1 mole equivalent of MTBE, 97%
Sinónimos:
(BrettPhos) palladium(II) phenethylamine chloride, BrettPhos Palladacycle, BrettPhos precatalyst, Chloro[2-(dicyclohexylphosphino)-3,6-dimethoxy-2′,4′, 6′-triisopropyl-1,1′-biphenyl][2-(2-aminoethyl)phenyl]palladium(II)
About This Item
Productos recomendados
Nivel de calidad
Ensayo
97%
Formulario
solid
Características
generation 1
idoneidad de la reacción
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
reaction type: Cross Couplings
mp
198-204 °C
grupo funcional
phosphine
cadena SMILES
COC(C)(C)C.NCCc1ccccc1[Pd]Cl.COc2ccc(OC)c(c2P(C3CCCCC3)C4CCCCC4)-c5c(cc(cc5C(C)C)C(C)C)C(C)C
InChI
1S/C35H53O2P.C8H10N.C5H12O.ClH.Pd/c1-23(2)26-21-29(24(3)4)33(30(22-26)25(5)6)34-31(36-7)19-20-32(37-8)35(34)38(27-15-11-9-12-16-27)28-17-13-10-14-18-28;9-7-6-8-4-2-1-3-5-8;1-5(2,3)6-4;;/h19-25,27-28H,9-18H2,1-8H3;1-4H,6-7,9H2;1-4H3;1H;/q;;;;+1/p-1
Clave InChI
OWHWOTGYDWMPCA-UHFFFAOYSA-M
Descripción general
Aplicación
Catalyst for:
- C,N-cross coupling of unprotected 3-halo-2-aminopyridines with primary and secondary amines
- Amination reaction
- N-arylation of aminophenols
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Contenido relacionado
The Buchwald group has developed a series of highly active and versatile palladium precatalysts and biarylphosphine ligands used in cross-coupling reactions for the formation of C-C, C–N, C–O, C–F, C–CF3, and C–S bonds. The ligands are electron-rich, and highly tunable to provide catalyst systems with a diverse scope, high stability and reactivity. Furthermore, the new series of precatalysts are air-, moisture and thermally-stable and display good solubility in common organic solvents. The use of precatalysts ensures the efficient generation of the active catalytic species and allows one to accurately adjust the ligand:palladium ratio. The ligands, precatalysts and methodology developed in the Buchwald group are user friendly and have rendered previously difficult cross couplings reactions, much easier to achieve.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico







 95%](/deepweb/assets/sigmaaldrich/product/structures/151/609/eeb99dc1-9ef2-49d8-b255-6b5e2519fee1/640/eeb99dc1-9ef2-49d8-b255-6b5e2519fee1.png)