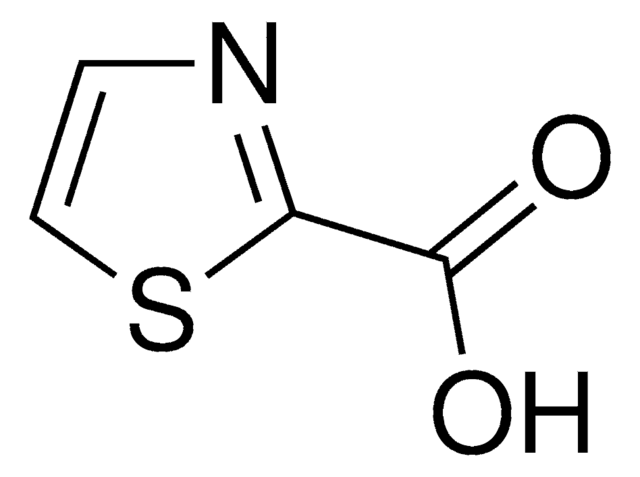
422460
2-Thiazolecarboxaldehyde
97%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C4H3NOS
Número de CAS:
Peso molecular:
113.14
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
assay
97%
form
liquid
refractive index
n20/D 1.574 (lit.)
bp
61-63 °C/15 mmHg (lit.)
density
1.288 g/mL at 25 °C (lit.)
functional group
aldehyde
storage temp.
2-8°C
SMILES string
O=Cc1nccs1
InChI
1S/C4H3NOS/c6-3-4-5-1-2-7-4/h1-3H
InChI key
ZGTFNNUASMWGTM-UHFFFAOYSA-N
General description
2-Thiazolecarboxaldehyde is a thiazole aldehyde derivative. It undergoes Baylis–Hillman reaction with methyl acrylate catalyzed by DABCO (1,4-diazabicyclo[2.2.2]octane). The reaction mechanism has been studied by electrospray ionization mass spectrometry (ESI-MS).
Application
2-Thiazolecarboxaldehyde may be used as a reactant in the following syntheses:
- Benzothiazine N-acylhydrazones, having potential antinociceptive and anti-inflammatory activity.
- Thiazole-2-yl-(amino)methylphosphonate diethyl esters.
- Imino ester by reacting with L-leucine t-butyl ester hydrochloride.
Useful building block for taxane analogs.
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
154.4 °F - closed cup
flash_point_c
68 °C - closed cup
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Armel A Agbodjan et al.
The Journal of organic chemistry, 73(8), 3094-3102 (2008-03-25)
A practical asymmetric synthesis of a highly substituted N-acylpyrrolidine on multi-kilogram scale is described. The key step in the construction of the three stereocenters is a [3+2] cycloaddition of methyl acrylate and an imino ester prepared from l-leucine t-butyl ester
The Morita-Baylis-Hillman Reaction: Advances and Contributions from Brazilian Chemistry.
Santos MS, et al.
Current Organic Synthesis, 12(6), 830-852 (2015)
Online mechanistic investigations of catalyzed reactions by electrospray ionization mass spectrometry: a tool to intercept transient species in solution.
Santos LS.
European Journal of Organic Chemistry, 2008(2), 235-253 (2008)
Yasuyuki Takeda et al.
Bioorganic & medicinal chemistry letters, 14(12), 3209-3215 (2004-05-20)
To improve the metabolic stability of 3, which exhibited both in vitro antitumor activity and in vivo efficacy by both iv and po administration, we designed and synthesized new taxane analogues. Most of the synthetic compounds maintained excellent antitumor activity
Synthesis of new thiazole-2,-4, and-5-yl-(amino) methylphosphonates and phosphinates: unprecedented cleavage of thiazole-2 derivatives under acidic conditions.
Olszewski TK and Boduszek B.
Tetrahedron, 66(45), 8661-8866 (2010)
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico





![Benzo[b]thiophene-2-carboxaldehyde 97%](/deepweb/assets/sigmaaldrich/product/structures/321/060/32405a4e-5720-4c6d-91cf-115c747270c4/640/32405a4e-5720-4c6d-91cf-115c747270c4.png)