214981
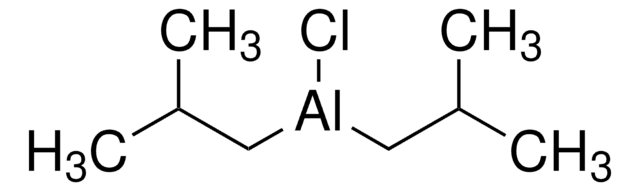
Hidruro de diisobutilaluminio solution
1.0 M in THF
Sinónimos:
DIBAL, DIBAL-H
About This Item
Productos recomendados
form
liquid
Quality Level
reaction suitability
reagent type: reductant
concentration
1.0 M in THF
bp
65 °C
density
0.866 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
CC(C)C[AlH]CC(C)C
InChI
1S/2C4H9.Al.H/c2*1-4(2)3;;/h2*4H,1H2,2-3H3;;
InChI key
AZWXAPCAJCYGIA-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
- Synthesis of trans-alkene isosteres of protected dipeptides.
- To generate bis(1,5-cyclooctadiene)nickel(0) (Ni(cod)2) in situ, which can catalyze the conjugate addition of ethenyltributyltin to 2-propenal to form tert-butyldimethyl[((E)-1,4pentadienyl)oxy]silane.
- Reduction of the arylpropiolate esters to give the corresponding propargyl alcohol.
Packaging
signalword
Danger
Hazard Classifications
Acute Tox. 4 Oral - Carc. 2 - Eye Dam. 1 - Flam. Liq. 2 - Pyr. Liq. 1 - Skin Corr. 1B - STOT SE 3 - Water-react 1
target_organs
Central nervous system, Respiratory system
supp_hazards
Storage Class
4.2 - Pyrophoric and self-heating hazardous materials
wgk_germany
WGK 1
flash_point_f
1.4 °F - closed cup
flash_point_c
-17 °C - closed cup
ppe
Faceshields, Gloves, Goggles
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico