161292
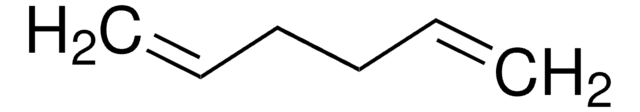
1,7-Octadiyne
98%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
HC≡C(CH2)4C≡CH
Número de CAS:
Peso molecular:
106.17
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
98%
form
liquid
refractive index
n20/D 1.446 (lit.)
bp
135-136 °C (lit.)
density
0.8 g/mL at 25 °C (lit.)
storage temp.
2-8°C
SMILES string
C#CCCCCC#C
InChI
1S/C8H10/c1-3-5-7-8-6-4-2/h1-2H,5-8H2
InChI key
DSOJWVLXZNRKCS-UHFFFAOYSA-N
Categorías relacionadas
General description
1,7-Octadiyne is a terminal bis-alkyne and participates in the formation of uranium(IV) vinyl complexes. It reacts with frustrated Lewis pair P(o-tolyl)3/B(C6F5)3 by acetylene C-C coupling to yield the zwitterionic product. It undergoes the reductive cyclization during reaction with Re2Cl4(μ-dppm)2 (dppm = Ph2PCH2PPh2) and affords quadruply bonded dirhenium(III) complex Re2Cl3(μ,η2-C8H7)(μ-dppm)2.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
flash_point_f
73.4 °F - closed cup
flash_point_c
23 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Chao Chen et al.
Chemical communications (Cambridge, England), 46(20), 3580-3582 (2010-04-09)
The frustrated Lewis pair P(o-tolyl)(3)/B(C(6)F(5))(3) reacts with 1,7-octadiyne by acetylene C-C coupling to yield the zwitterionic product 2a. In contrast, the P(o-tolyl)(3)/B(C(6)F(5))(3) Lewis pair reacts with 1,6-heptadiyne by a sequence involving 1,1-carboboration of a terminal acetylene to eventually yield the
A Novel Example of the Reductive Cyclization of a Diyne at a Re-Re Triple Bond: The Reaction of Re2Cl4 (?-dppm) 2 with 1, 7-Octadiyne.
Ganesan M, et al.
Organometallics, 22(4), 870-872 (2003)
Boris Kosog et al.
Journal of the American Chemical Society, 134(30), 12792-12797 (2012-06-29)
The previously reported uranium(III) complex [(((Ad)ArO)(3)N)U(III)(DME)] (1; Ad = adamantane, DME = 1,2-dimethoxyethane) reacts with the terminal bis-alkynes 1,7-octadiyne or 1,6-heptadiyne in C-C-coupling reactions to form the uranium(IV) vinyl complexes [{(((Ad)ArO)(3)N)U(IV)}(2)(μ-η(2):η(1)-1,2-(CH)(2)-cyclohexane)] (2) and [{(((Ad)ArO)(3)N)U(IV)}(2)(μ-η(2):η(2)-1,2-(CH)(2)-cyclopentane)] (3). With the monoalkynes 1-hexyne or
Christina A Braun et al.
Dalton transactions (Cambridge, England : 2003), 48(27), 10210-10219 (2019-06-14)
A new di(isopropoxy)boryl -B(OiPr)2 tellurophene precursor is described, from which several previously inaccessible phosphorescent borylated tellurophenes are formed via exchange of the -OiPr groups. One such tellurophene Mes(iPrO)B-Te-6-B(OiPr)Mes, bearing a sterically encumbered mesityl (Mes) substituent at each boron center, exhibits
Shasha Wang et al.
Scientific reports, 7(1), 12712-12712 (2017-10-07)
We developed an efficient one-pot metal-free click polymerization procedure for the synthesis of 3,5-disubstituted polypyrazoles with high yields, high molecular weights, and narrow molecular weight distribution. The method involved two click reactions in a one-pot synthesis. The first reaction was
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico