159530
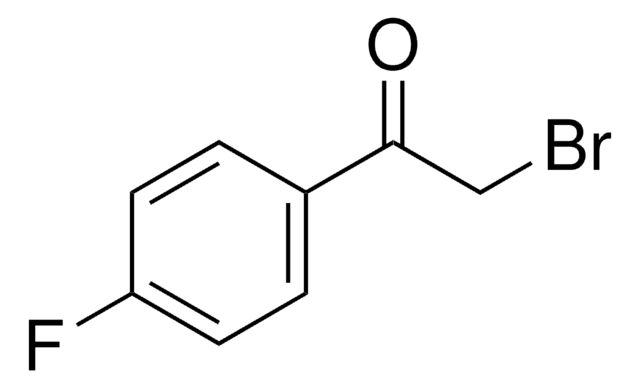
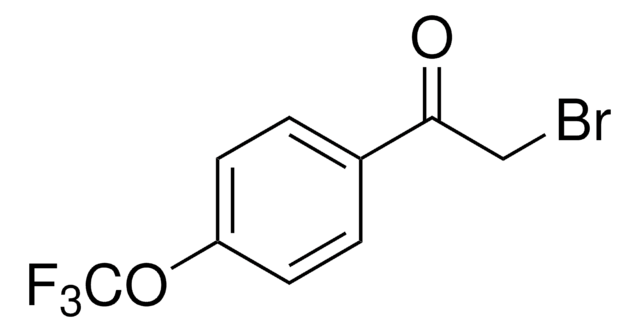
2-Bromo-4′-methylacetophenone
90%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
CH3C6H4COCH2Br
Número de CAS:
Peso molecular:
213.07
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
assay
90%
bp
105 °C/0.1 mmHg (lit.)
mp
45-49 °C (lit.)
functional group
bromo
ketone
storage temp.
2-8°C
SMILES string
Cc1ccc(cc1)C(=O)CBr
InChI
1S/C9H9BrO/c1-7-2-4-8(5-3-7)9(11)6-10/h2-5H,6H2,1H3
InChI key
KRVGXFREOJHJAX-UHFFFAOYSA-N
General description
2-Bromo-4′-methylacetophenone is an α-bromoketone.
Application
2-Bromo-4′-methylacetophenone was used in the general fluorous thiol quenching method. It was also used in the preparation of hydroxyquinolinone and N-derivatized carboxamides.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
235.4 °F - closed cup
flash_point_c
113 °C - closed cup
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Use of fluorous silica gel to separate fluorous thiol quenching derivatives in solution-phase parallel synthesis
Zhang W, et al.
Tetrahedron, 58(20), 3871-3875 (2002)
Miroslav Soural et al.
Journal of combinatorial chemistry, 9(5), 793-796 (2007-08-07)
A highly efficient solid-phase synthesis of 2-substituted-3-hydroxy-4(1H)-quinolinone-7-carboxamides was developed using anthranilates and bromoketones as the key synthons. Primary amines immobilized to an acid-cleavable BAL linker were acylated with 1-methyl-2-aminoterephtalate. After cleavage of the methyl ester, bromoketones were used to form
Hana Elshaflu et al.
Frontiers in chemistry, 6, 247-247 (2018-07-19)
The novel approach in the treatment of complex multifactorial diseases, such as neurodegenerative disorders and cancer, requires a development of efficient multi-targeting oriented drugs. Since oxidative stress significantly contributes to the pathogenesis of cancer and neurodegenerative disorders, potential drug candidates
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico