156329
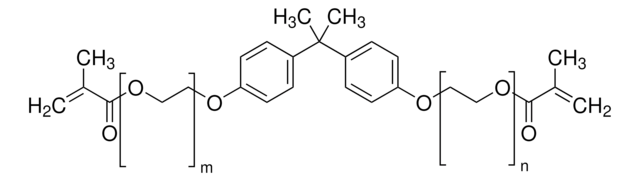
Bisphenol A dimethacrylate
>98%
Sinónimos:
2,2-Bis(4-hydroxyphenyl)propane dimethacrylate, 2,2-Bis(4-methacryloxyphenyl)propane, 2,2-Bis(4-methacryloyloxyphenyl)propane, 4,4′-Isopropylidenediphenol dimethacrylate, BPADMA
About This Item
Productos recomendados
Ensayo
>98%
Formulario
solid
mp
72-74 °C (lit.)
temp. de almacenamiento
2-8°C
cadena SMILES
CC(=C)C(=O)Oc1ccc(cc1)C(C)(C)c2ccc(OC(=O)C(C)=C)cc2
InChI
1S/C23H24O4/c1-15(2)21(24)26-19-11-7-17(8-12-19)23(5,6)18-9-13-20(14-10-18)27-22(25)16(3)4/h7-14H,1,3H2,2,4-6H3
Clave InChI
QUZSUMLPWDHKCJ-UHFFFAOYSA-N
Categorías relacionadas
Descripción general
Aplicación
- A template in the synthesis of molecularly imprinted polymers (MIPs). The use of BPA-DMA allows for the formation of specific cavities in the polymer matrix that match the shape and functional groups of BPA, enhancing the selectivity and affinity of the resulting polymer for its target analyte.
- A co-monomer in the synthesis of photopolymerized monoliths for capillary electrochromatography.
- A key component in the production of bisphenol A-glycidyl methacrylate (Bis-GMA), which is widely used in dental restorative materials due to its mechanical properties and potential for additional functionalities like antibacterial activity.
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico