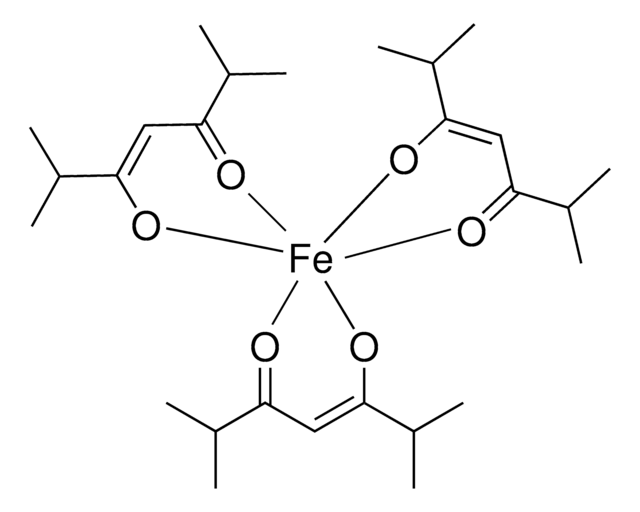
278505
Sulfuryl chloride solution
1.0 M in methylene chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
SO2Cl2
CAS Number:
Molecular Weight:
134.97
MDL number:
UNSPSC Code:
12352300
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
form
liquid
Quality Level
concentration
1.0 M in methylene chloride
density
1.352 g/mL at 25 °C
SMILES string
ClS(Cl)(=O)=O
InChI
1S/Cl2O2S/c1-5(2,3)4
InChI key
YBBRCQOCSYXUOC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Palladium-Catalyzed Synthesis of Ammonium Sulfinates from Aryl Halides and a Sulfur Dioxide Surrogate: A Gas-and Reductant-Free Process: This study elaborates on using sulfuryl chloride for oxidative chlorination leading to sulfonyl chloride formation, which is important for synthetic organic chemistry (Emmett et al., 2014).
- Combining Organometallic Reagents, the Sulfur Dioxide Surrogate DABSO, and Amines: A One‐Pot Preparation of Sulfonamides, Amenable to Array Synthesis: This research discusses an innovative one-pot method for preparing sulfonamides using sulfuryl chloride, highlighting its utility in streamlining complex syntheses (Deeming et al., 2015).
- Interfacial polymerization: from chemistry to functional materials: The article illustrates the use of sulfuryl chloride in interfacial polymerization, a key technique for developing advanced materials with tailored properties (Zhang et al., 2020).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Inhalation - Carc. 2 - Eye Dam. 1 - Skin Corr. 1C - STOT SE 3
Target Organs
Central nervous system
Supplementary Hazards
Storage Class Code
6.1B - Non-combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Zhiyong Wu et al.
Organic letters, 15(6), 1270-1273 (2013-03-07)
An efficient and concise one-pot protocol to synthesize sulfonylated quinoline N-oxides via copper-catalyzed C-H bond activation has been developed. Commercially available and less expensive aryl sulfonyl chlorides were used as the sulfonylation reagents. Various 2-aryl sulfonyl quinolines were obtained in
Christopher Blackburn
ACS combinatorial science, 14(3), 150-154 (2012-02-11)
Reductive aminations and further transformations of an azo dye and fluorous tagged aldehyde are described. The intensely colored 2,4-dialkoxybenzyl protected amines undergo Fmoc-based peptide coupling, Suzuki reactions, and sulfonamide formation with product isolation facilitated by visual monitoring of fluorous solid
Polystyrene sulfonyl chloride: a highly orthogonal linker resin for the synthesis of nitrogen-containing heterocycles.
Matthias Mentel et al.
Angewandte Chemie (International ed. in English), 48(32), 5841-5844 (2009-07-08)
Iron-catalyzed desulfinylative C-C cross-coupling reactions of sulfonyl chlorides with grignard reagents.
Chandra M Rao Volla et al.
Angewandte Chemie (International ed. in English), 47(7), 1305-1307 (2008-01-05)
Vlad Martin-Diaconescu et al.
Inorganic chemistry, 48(3), 1038-1044 (2009-01-10)
The electronic structure of organic sulfonyl compounds of the form RSO(2)G (G = -Cl, -OH, -CH(3)) is investigated to evaluate the effect of aryl R groups on photocleavage of the S-G bond. Sulfur K-edge X-ray absorption spectroscopy (XAS) provides a
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service