推荐产品
化驗
≥98% (HPLC)
形狀
solid
藥物控制
USDEA Schedule IV; Home Office Schedule 3; stupéfiant (France); kontrollierte Droge in Deutschland; regulated under CDSA - not available from Sigma-Aldrich Canada; psicótropo (Spain); Decreto Lei 15/93: Tabela IIC (Portugal)
儲存條件
protect from light
顏色
white
溶解度
DMSO: >10 mg/mL
起源
Sanofi Aventis
儲存溫度
room temp
SMILES 字串
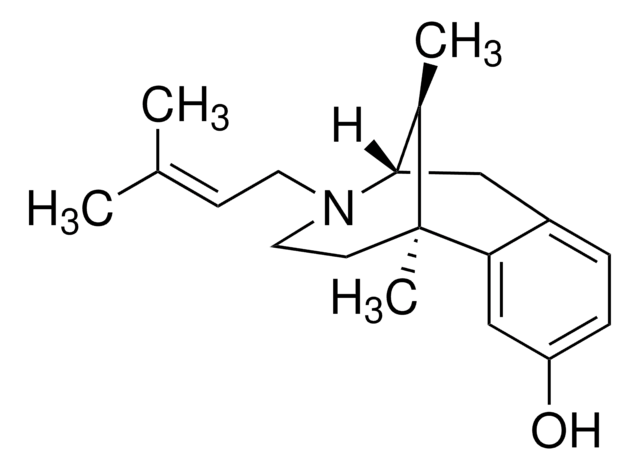
C[C@@H]1[C@@H]2Cc3ccc(O)cc3[C@@]1(C)CCN2C\C=C(\C)C
InChI
1S/C19H27NO/c1-13(2)7-9-20-10-8-19(4)14(3)18(20)11-15-5-6-16(21)12-17(15)19/h5-7,12,14,18,21H,8-11H2,1-4H3/t14-,18+,19+/m1/s1
InChI 密鑰
VOKSWYLNZZRQPF-CCKFTAQKSA-N
生化/生理作用
Pentazocine is an analgesic with both agonist and antagonist activities. It acts as a κ- and σ-opioid receptor agonist and partial mu-receptor antagonist. Pentazocine pre-treatment reduces the frequency of fentanyl-induced cough and improves the anesthetic effect.
特點和優勢
This compound was developed by Sanofi Aventis. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Oral
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
Ralph Holl et al.
Journal of medicinal chemistry, 52(7), 2126-2137 (2009-02-27)
A series of 6,8-diazabicyclo[3.2.2]nonane derivatives bearing two aromatic moieties was prepared, the affinity toward sigma(1) and sigma(2) receptors was investigated, and the growth inhibition of six human tumor cell lines was determined. The enantiopure bicyclic ketones 5a ((+)-(1S,5S)-6-allyl-8-(4-methoxybenzyl)-6,8-diazabicyclo[3.2.2]nonane-2,7,9-trione) and 5b
Ling-Wei Hsin et al.
Journal of medicinal chemistry, 53(3), 1392-1396 (2010-01-09)
A series of enantiomeric N-substituted 2,3,4,4a,5,6,7,7a-octahydro-1H-benzofuro[3,2-e]isoquinolines was synthesized. The (-)-enantiomers had much greater kappa-, mu-, and delta-opioid receptor binding affinity than the corresponding (+)-enantiomers. Compounds (-)-1a, (-)-1b, and (-)-1c displayed subnanomolar binding affinity for the mu-receptor, and (-)-1b had a
Eva Grosse Maestrup et al.
Bioorganic & medicinal chemistry, 19(1), 393-405 (2010-12-04)
In order to develop a fluorinated radiotracer for imaging of σ(1) receptors in the central nervous system a series of (2-fluoroethyl) substituted spirocyclic piperidines 3 has been prepared. In the key step of the synthesis 2-bromocinnamaldehyde acetal 5 was added
Christian Geiger et al.
Journal of medicinal chemistry, 50(24), 6144-6153 (2007-10-31)
All possible stereoisomeric alcohols (6-benzyl-8-(4-methoxybenzyl)-6,8-diazabicyclo[3.2.2]nonan-2-ol) and methyl ethers (6-benzyl-2-methoxy-8-(4-methoxybenzyl)-6,8-diazabicyclo[3.2.2]nonane) are prepared from (R)- and (S)-glutamate. A Dieckmann analogous cyclization, which makes use of trapping the primary cyclization product with Me3SiCl, generates the bicyclic framework. Stereoselective LiBH4 reduction and Mitsunobu inversion
Carmen Abate et al.
Journal of medicinal chemistry, 54(4), 1022-1032 (2011-01-15)
1-Cyclohexyl-4-[3-(5-methoxy-1,2,3,4-tetrahydronaphthalen-1-yl)propyl]piperazine 1 (PB28) represents an excellent lead candidate for therapeutic and/or diagnostic applications in oncology. However, because its utility is limited by its relatively high degree of lipophilicity, novel analogues of 1 with reduced lipophilic character were designed by substituting
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门