推荐产品
等級
pharmaceutical primary standard
API 家族
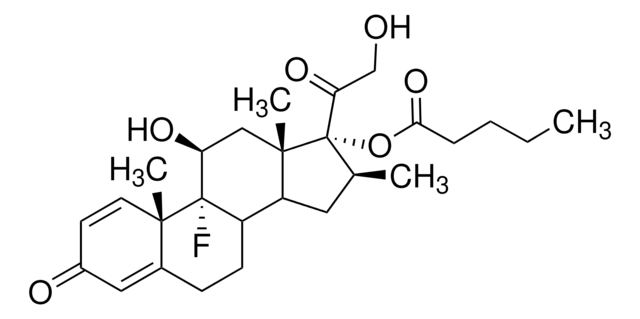
betamethasone
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-8°C
SMILES 字串
CCCCC(=O)O[C@@]1([C@@H](C)CC2C3CCC4=CC(=O)C=C[C@]4(C)[C@@]3(F)[C@@H](O)C[C@]12C)C(=O)CO
InChI
1S/C27H37FO6/c1-5-6-7-23(33)34-27(22(32)15-29)16(2)12-20-19-9-8-17-13-18(30)10-11-24(17,3)26(19,28)21(31)14-25(20,27)4/h10-11,13,16,19-21,29,31H,5-9,12,14-15H2,1-4H3/t16-,19?,20?,21-,24-,25-,26-,27-/m0/s1
InChI 密鑰
SNHRLVCMMWUAJD-QDHNOTTGSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Betamethasone valerate for system suitability EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包裝
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他說明
Sales restrictions may apply.
相關產品
产品编号
说明
价格
訊號詞
Danger
危險聲明
危險分類
Repr. 1B - STOT RE 2
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 2
Christine Bangert et al.
Dermatology (Basel, Switzerland), 222(1), 36-48 (2010-12-15)
Topical pimecrolimus may maintain remissions of atopic dermatitis (AD) by inhibiting subclinical inflammation. To evaluate clinical and cytological effects of pimecrolimus in topical corticosteroid-treated and resolved AD lesions. Patients (n=67) with resolved AD lesions were randomized to 3-week double-blind treatment
Prakash Adhikari et al.
International journal of pediatric otorhinolaryngology, 75(4), 500-503 (2011-02-05)
This study was carried out with the objective of comparing clinical efficacy of 10% ichthammol glycerine (IG) pack with steroid-antibiotic pack for relieving pain in cases of acute otitis externa. A prospective quasi-randomized clinical trial was completely carried out in
Saif-ur-Rehman Khattak et al.
AAPS PharmSciTech, 14(1), 177-182 (2012-12-20)
The effects of solvent [acetonitrile, methanol, and acetonitrile/water mixture (20:80, v/v)], buffer concentration (phosphate buffer, pH 7.5), ionic strength and commonly employed adjuvants on the photodegradation of betamethasone-17 valerate in cream and gel formulations have been studied on exposure to UV
Jens-Michael Jensen et al.
Experimental dermatology, 20(10), 783-788 (2011-06-29)
It has been suggested that the increased rate of bacterial infection in atopic dermatitis (AD) may be caused by reduced antimicrobial protein (AMP) expression. We were interested whether common treatments in AD affect antimicrobial defense. We investigated the effects of
Annabel Maruani et al.
International journal of pharmaceutics, 395(1-2), 84-90 (2010-05-18)
Topical steroids are efficient in vasoconstriction potential, which is linked to their anti-inflammatory activity. Low-frequency ultrasound (US) applied on the skin (sonophoresis) may enhance the transdermal transport of various steroids. We aimed to assess, in a simple, blinded, randomized controlled
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门