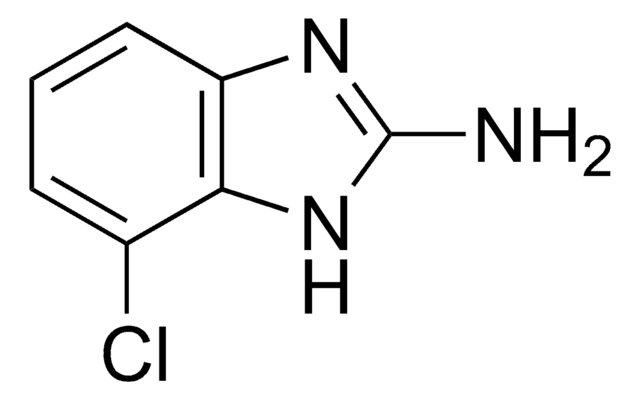
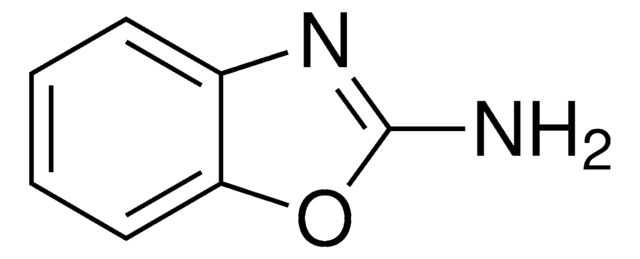
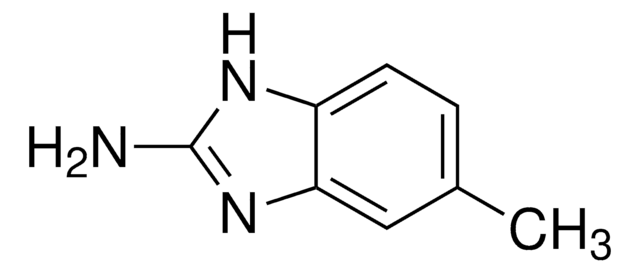
推荐产品
等级
analytical standard
质量水平
产品线
PESTANAL®
保质期
limited shelf life, expiry date on the label
技术
HPLC: suitable
gas chromatography (GC): suitable
mp
226-230 °C (lit.)
应用
agriculture
environmental
包装形式
neat
SMILES字符串
Nc1nc2ccccc2[nH]1
InChI
1S/C7H7N3/c8-7-9-5-3-1-2-4-6(5)10-7/h1-4H,(H3,8,9,10)
InChI key
JWYUFVNJZUSCSM-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
应用
Refer to the product′s Certificate of Analysis for more information on a suitable instrument technique. Contact Technical Service for further support.
推荐产品
Find a digital Reference Material for this product available on our online platform ChemisTwin® for NMR. You can use this digital equivalent on ChemisTwin® for your sample identity confirmation and compound quantification (with digital external standard). An NMR spectrum of this substance can be viewed and an online comparison against your sample can be performed with a few mouseclicks. Learn more here and start your free trial.
法律信息
PESTANAL is a registered trademark of Merck KGaA, Darmstadt, Germany
警示用语:
Warning
危险声明
危险分类
Acute Tox. 4 Oral
储存分类代码
11 - Combustible Solids
WGK
WGK 3
个人防护装备
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Min Zhong et al.
Bioorganic & medicinal chemistry letters, 19(17), 5158-5161 (2009-08-04)
This Letter describes the discovery and key structure-activity relationship (SAR) of a series of 2-aminobenzimidazoles as potent Aurora kinase inhibitors. 2-Aminobenzimidazole serves as a bioisostere of the biaryl urea residue of SNS-314 (1c), which is a potent Aurora kinase inhibitor
Ahmad Shaabani et al.
Combinatorial chemistry & high throughput screening, 9(10), 771-776 (2006-12-16)
Tetraheterocyclic benzimidazo[1,2-a]quinazolin-4(1H)-one and tetrahydro-1,2,4-triazolo[5,1-b]quinazolin-8(4H)-one were synthesized in relatively high yields by the condensation reaction of an aldehyde and a cyclic beta-diketone with 2-aminobenzothiazole, 2-aminobenzimidazole or 3-amino-1,2,4-triazole without using any catalyst under solvent-free conditions.
Zinc fingered: new compounds that thwart gram-positive biofilm formation by sequestering zinc.
Dinty J Musk
Chembiochem : a European journal of chemical biology, 11(6), 758-760 (2010-03-02)
2-Aminobenzimidazole derivatives strongly inhibit and disperse Pseudomonas aeruginosa biofilms.
Reto Frei et al.
Angewandte Chemie (International ed. in English), 51(21), 5226-5229 (2012-04-11)
S Sudha et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 84(1), 184-195 (2011-10-11)
In the present work, we reported a combined experimental and theoretical study on molecular structure, vibrational spectra and HOMO-LUMO analysis of 2-aminobenzimidazole (2-ABD). The FTIR (400-4000 cm(-1)) and FT-Raman spectra (50-3500 cm(-1)) of 2-ABD were recorded. The molecular geometry, harmonic
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门