所有图片(1)
About This Item
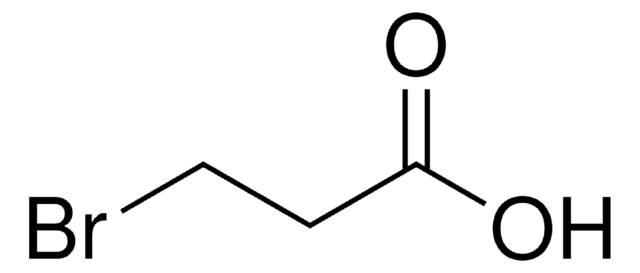
线性分子式:
H2NC4H6CO2H
CAS号:
分子量:
115.13
MDL编号:
UNSPSC代码:
12352106
PubChem化学物质编号:
NACRES:
NA.22
推荐产品
质量水平
方案
97%
表单
solid
反应适用性
reaction type: solution phase peptide synthesis
mp
261 °C (dec.) (lit.)
应用
peptide synthesis
SMILES字符串
NC1(CCC1)C(O)=O
InChI
1S/C5H9NO2/c6-5(4(7)8)2-1-3-5/h1-3,6H2,(H,7,8)
InChI key
FVTVMQPGKVHSEY-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
警示用语:
Warning
危险分类
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
靶器官
Respiratory system
储存分类代码
11 - Combustible Solids
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
个人防护装备
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
1-Aminocyclobutane-1-carboxylate (ACBC): a specific antagonist of the N-methyl-D-aspartate receptor coupled glycine receptor.
W F Hood et al.
European journal of pharmacology, 161(2-3), 281-282 (1989-02-28)
Insulin-induced phospho-oligosaccharide stimulates amino acid transport in isolated rat hepatocytes.
I Varela et al.
The Biochemical journal, 267(2), 541-544 (1990-04-15)
The ability of the insulin-induced phospho-oligosaccharide to stimulate amino acid transport was studied in isolated rat hepatocytes. At low alpha-aminoisobutyric acid concentrations (0.1 mM), both 100 nM-insulin and 10 microM-phospho-oligosaccharide doubled amino acid uptake after 2 h of incubation. This
Y Gaoni et al.
Journal of medicinal chemistry, 37(25), 4288-4296 (1994-12-09)
A range of cis- and trans-3-substituted 1-aminocyclobutane-1-carboxylic acids has been synthesized and evaluated for antagonism at excitatory amino acid receptor sites and for anticonvulsant activity. Potent and selective antagonist activity at N-methyl-D-aspartate (NMDA) receptor sites in neonatal rat motoneurones was
J K Relton et al.
Brain research bulletin, 32(3), 223-226 (1993-01-01)
Arachidonic acid [20:4(N-6)] has been implicated in neurological damage induced by cerebral ischaemia. Membrane arachidonate concentrations can be reduced by changes in dietary fat intake. Therefore, in the present study, we have investigated the effects of N-3 fatty acid supplementation
M C Pirrung et al.
Chemistry & biology, 5(1), 49-57 (1998-02-28)
The chemical mechanism of the final step of ethylene biosynthesis (the conversion of 1-aminocyclopropanecarboxylic acid, ACC, to ethylene by ACC oxidase, the ethylene-forming enzyme, EFE) is poorly understood. Two possibilities have been suggested: a radical mechanism and an N-hydroxylation mechanism.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持