所有图片(1)
About This Item
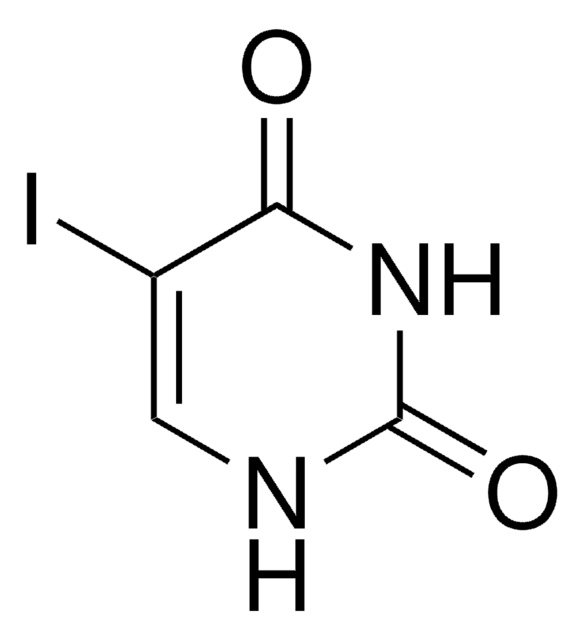
经验公式(希尔记法):
C7H10N2O3
CAS号:
分子量:
170.17
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推荐产品
化驗
99%
mp
51-54 °C (lit.)
SMILES 字串
COc1cc(OC)nc(OC)n1
InChI
1S/C7H10N2O3/c1-10-5-4-6(11-2)9-7(8-5)12-3/h4H,1-3H3
InChI 密鑰
RJVAFLZWVUIBOU-UHFFFAOYSA-N
一般說明
2,4,6-Trimethoxypyrimidine is a pyrimidine derivative. One of the methods reported for its synthesis is by the reaction of 2,4,6-trichloropyrimidine with sodium ethoxide at 70-100°C. Its transformation into 1,6-dihydro-2,4-dimethoxy-1-methyl-6-oxopyrimidine by Hilbert-Johnson reaction has been reported.
應用
2,4,6-Trimethoxypyrimidine may be used as a model compound in a study to determine the qualitative composition of mixtures formed during the methylation of barbituric acid and its derivatives by diazomethane.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
Solvation effects in the methylation of barbituric acid and its derivatives by diazomethane.
Krasnov KA, et al.
Chemistry of Heterocyclic Compounds, 23(11), 1218-1221 (1987)
Chemistry of heterocyclic compounds. 29. Synthesis and reactions of multihetero macrocycles possessing 2, 4-pyrimidino subunits connected by carbon-oxygen and/or-sulfur linkages.
Newkome GR, et al.
The Journal of Organic Chemistry, 43(17), 3362-3367 (1978)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门