Q4951
Quercetin
≥95% (HPLC), solid, anticancer agent
Sinónimos:
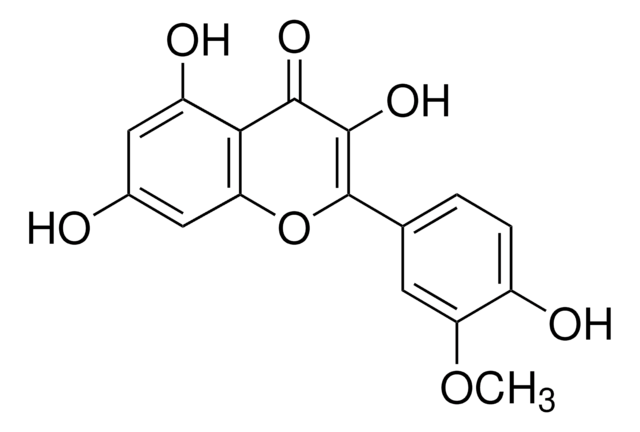
2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-4H-1-benzopyran-4-one, 3,3′,4′,5,6-Pentahydroxyflavone
About This Item
Productos recomendados
product name
Quercetin, ≥95% (HPLC), solid
biological source
synthetic (organic)
assay
≥95% (HPLC)
form
solid
mp
316.5 °C
solubility
water: practically insoluble
storage temp.
room temp
SMILES string
OC1=CC(O)=C2C(OC(C3=CC=C(O)C(O)=C3)=C(O)C2=O)=C1
InChI
1S/C15H10O7/c16-7-4-10(19)12-11(5-7)22-15(14(21)13(12)20)6-1-2-8(17)9(18)3-6/h1-5,16-19,21H
InChI key
REFJWTPEDVJJIY-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
- Quercetin has been used as an antioxidant which reversed the immunosuppressive effects of high glucose and hyperglycemic sera in type 2 diabetic patients.
- It has been used as a detoxifying phytochemical in Apis mellifera.
- It has been used as a positive control in DPPH (2,2- diphenyl-1-picryhydrazyl) radical scavenging assay. It has also been used for the preparation of calibration curve to determine total flavonoid content.
Biochem/physiol Actions
Features and Benefits
Preparation Note
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
ppe
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Fatty acid synthesis supports cancer cell proliferation, essential for membrane generation, protein modification, and bioenergetics.
Fatty acid synthesis supports cancer cell proliferation, essential for membrane generation, protein modification, and bioenergetics.
Fatty acid synthesis supports cancer cell proliferation, essential for membrane generation, protein modification, and bioenergetics.
Fatty acid synthesis supports cancer cell proliferation, essential for membrane generation, protein modification, and bioenergetics.
Protocolos
Protocol for HPLC Analysis of Flavonoids on Ascentis® RP-Amide
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico