362069
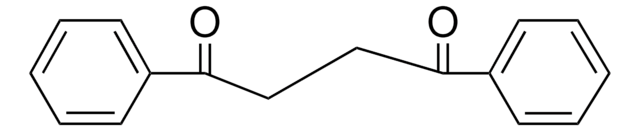
1,3-Dibenzoylpropane
98%
Synonym(s):
1,5-Diphenyl-1,5-pentanedione
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
C6H5CO(CH2)3COC6H5
CAS Number:
Molecular Weight:
252.31
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
solid
mp
66-68 °C (lit.)
SMILES string
O=C(CCCC(=O)c1ccccc1)c2ccccc2
InChI
1S/C17H16O2/c18-16(14-8-3-1-4-9-14)12-7-13-17(19)15-10-5-2-6-11-15/h1-6,8-11H,7,12-13H2
InChI key
YOLLTWVIOASMFW-UHFFFAOYSA-N
Related Categories
General description
1,3-Dibenzoylpropane is a 1,3-diaroylpropane. 1,3-Dibenzoylpropane is formed during hydroxocobalt(III) Schiff base complexes catalyzed selective aldol reaction of dibenzoylmethanes with formaldehyde in methanol.
Application
1,3-Dibenzoylpropane may be used in the electrochemical synthesis of cis-1,2-diphenyl-1,2-cyclopentanediol, via reduction in acetonitrile.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Electrochemical cyclization: II. Intramolecular pinacolization of 1, 3-dibenzoylpropane in acetonitrile.
Ammar F, et al.
J. Electroanal. Chem. Interfac. Electrochem., 53(3), 407-416 (1974)
Highly selective aldol reaction of dibenzoylmethanes with formaldehyde catalyzed by cobalt Schiff base complex under neutral conditions.
Maruyama K, et al.
Tetrahedron Letters, 36(31), 5609-5612 (1995)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service




![2,3,6,7-Tetrahydro-8-hydroxy-1H,5H-benzo[ij]quinolizine-9-carboxaldehyde 98%](/deepweb/assets/sigmaaldrich/product/structures/166/830/a0d9a84a-5623-41a1-a54b-3b0272e5b28c/640/a0d9a84a-5623-41a1-a54b-3b0272e5b28c.png)