926175
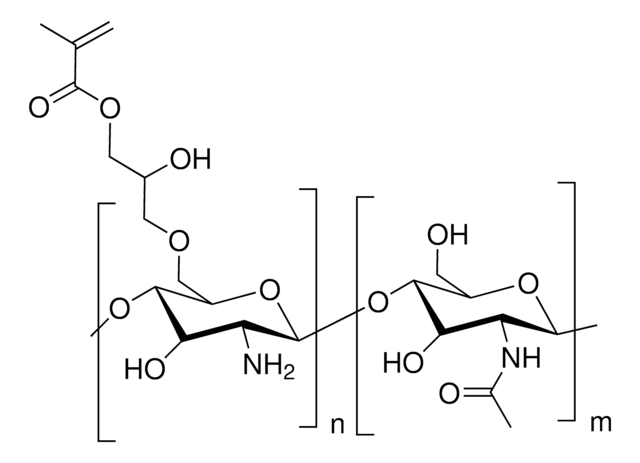
Glycol Chitosan Methacrylate
Degree of methacrylation ∼45%
Synonyme(s) :
Chitosan hydrogel, Crosslinkble chitosan, Glycol chitosan, Methacrylated glycol chitosan
About This Item
Produits recommandés
Niveau de qualité
Forme
powder
Couleur
white to light yellow, light brown
Adéquation
conforms to structure for NMR
Température de stockage
2-8°C
Application
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
Vous ne trouvez pas la bonne version ?
Si vous avez besoin d'une version particulière, vous pouvez rechercher un certificat spécifique par le numéro de lot.
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique