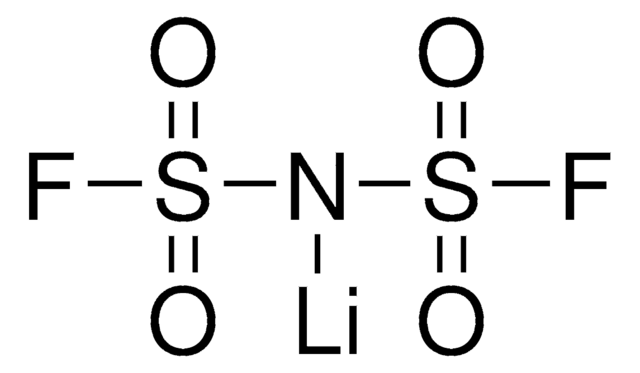919977
Bis(trifluorométhane)sulfonimide lithium salt
anhydrous, 99.99% trace metals basis
Synonyme(s) :
Bis(trifluorométhylsulfonyl)amine lithium salt, Bistrifluorométhanesulfonimidate de lithium
About This Item
Produits recommandés
Qualité
anhydrous
Niveau de qualité
Pureté
99.99% trace metals basis
Caractéristiques du produit alternatif plus écologique
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
Pf
234-238 °C (lit.)
Application(s)
battery manufacturing
Autre catégorie plus écologique
Chaîne SMILES
[Li]N(S(=O)(=O)C(F)(F)F)S(=O)(=O)C(F)(F)F
InChI
1S/C2F6NO4S2.Li/c3-1(4,5)14(10,11)9-15(12,13)2(6,7)8;/q-1;+1
Clé InChI
QSZMZKBZAYQGRS-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- An additive in the development of dual-functional separator coating materials. These materials are based on covalent organic frameworks (COFs) and are specifically designed for use in high-performance lithium-selenium sulfide batteries. The Li-SeS2 battery achieved outstanding performance in terms of energy storage and stability. It exhibited a specific capacity of 844.6 mA h g-1 at 0.5C and a SeS2 loading of 2 mg cm-2.
- As an additive in the electrolyte formulation along with polyethylene oxide for the development of solid-state lithium batteries. LiTFSI enhance the ionic conductivity of the PEO-based electrolyte, which is essential for the efficient transport of lithium ions.
- As a key component in the development of a PEO/LiTFSI-coated polypropylene membrane. This membrane is designed for high-loading lithium–sulfur batteries to enhance battery performance, improve capacity, and extend cycle life.
- As a component in the electrolyte system along with TEMPOL derivatives. The incorporation of LiTFSI in the electrolyte system enhances the stability and achieves an efficiency of 6.16% in solid-state fiber dye-sensitized solar cells.
Produit(s) apparenté(s)
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B - STOT RE 2 Oral
Organes cibles
Nervous system
Code de la classe de stockage
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
Vous ne trouvez pas la bonne version ?
Si vous avez besoin d'une version particulière, vous pouvez rechercher un certificat spécifique par le numéro de lot.
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique