901546
Poly(M(EO)2MA:Poly(OEGMA) 90:10
hydrazide functionalized, 25 wt. % (solution in water)
Synonyme(s) :
PEGMA-co-POEGMA
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
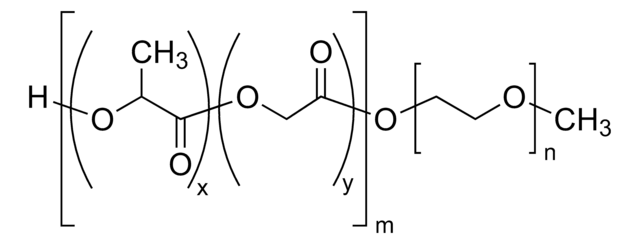
Formule linéaire :
[CH2CCH3(COO(CH2CH2O)2CH3)]p[CH2CCH3(COO(CH2CH2O)8CH3)]n[CH2CHCONHNHCO(CH2)4CONHNH2]m
Code UNSPSC :
12162002
Nomenclature NACRES :
NA.23
Produits recommandés
Forme
solution
Poids mol.
Mw ~20,000 g/mol
Concentration
25 wt. % (solution in water)
Couleur
clear colorless to pale yellow
Groupe fonctionnel
hydrazide
Température de stockage
2-8°C
Application
In addition, the physical properties of the resulting hydrogels, such as LCST, gelation rates, swelling kinetics, degradation kinetics, and mechanical properties, can all be readily controlled by solution concentration and the ratios of each solution.
This product is provided as a 25 wt% solution in water, ready to be diluted for your specific application. Please see the technical bulletin on the product page for dilution instructions and hydrogel preparation instructions.
This product is provided as a 25 wt% solution in water, ready to be diluted for your specific application. Please see the technical bulletin on the product page for dilution instructions and hydrogel preparation instructions.
Poly(diethylene glycol methacrylate)-co-(oligoethylene glycol methacrylate), (PM(EO)2MA-co-OEGMA) is a comb-shaped, graft copolymer consisting of hydrophilic oligomer polyethylene glycol (PEG) chains grafted to a hydrophobic polymethacrylate backbone. In this material, the PEG chains are either diethylene glycol units or oligomer PEG (n=8-9) units. Resulting material properties can be tuned by controlling the ratio of each component. POEGMA has been suggested as a viable alternative to PEG in biological and biomaterial applications. POEGMA has been reported to improve pharmacokinetic properties of protein and peptide conjugates, enhance the stability and gene silencing efficiency of siRNAs, as an anti-fouling surface for biosensors, and eliminate PEG antigenicity. In addition to use in biomolecule-polymer conjugates, PEOGMA has also seen wide spread use in tissue engineering applications, such as hydrogel synthesis. Hydrazide-functionalized PM(EO)2MA-co-OEGMA can be readily used with the corresponding aldehyde-functionalized POEGMA or PM(EO)2MA-co-OEGMA for rapid gelation via reversible hydrazone bond formation. Due to the reversibility of the bond formation and the low viscosity of the precursors, resulting hydrogels can be used as injectable tissue engineering matrices, local drug delivery vehicles for small molecules, or as joint lubricants.
Code de la classe de stockage
10 - Combustible liquids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
Lot/Batch Number
Vous ne trouvez pas la bonne version ?
Si vous avez besoin d'une version particulière, vous pouvez rechercher un certificat spécifique par le numéro de lot.
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
A brush-polymer/exendin-4 conjugate reduces blood glucose levels for up to five days and eliminates poly(ethylene glycol) antigenicity.
Qi Y, et al.
Nature Biomedical Engineering, 1, 0002-0002 (2016)
Reactive electrospinning of degradable poly(oligoethylene glycol methacrylate)-based nanofibrous hydrogel networks.
Xu F, et al.
Chemical Communications (Cambridge, England), 52, 1451-1454 (2016)
Injectable hydrogels based on poly(ethylene glycol) and derivatives as functional biomaterials.
Bakaic E, et al.
Royal Society of Chemistry Advances, 5, 35469-35486 (2015)
?Off-the-shelf? thermoresponsive hydrogel design: tuning hydrogel properties by mixing precursor polymers with different lower-critical solution temperatures.
Bakaic E, et al.
Royal Society of Chemistry Advances, 5, 33364-33376 (2015)
Weiping Gao et al.
Proceedings of the National Academy of Sciences of the United States of America, 106(36), 15231-15236 (2009-08-27)
The challenge in the synthesis of protein-polymer conjugates for biological applications is to synthesize a stoichiometric (typically 1:1) conjugate of the protein with a monodisperse polymer, with good retention of protein activity, significantly improved pharmacokinetics and increased bioavailability, and hence
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique