799718
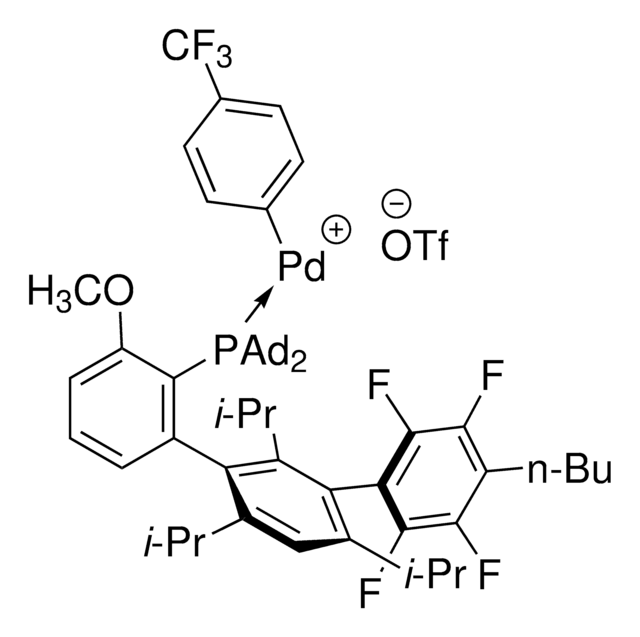
AlPhos
Synonyme(s) :
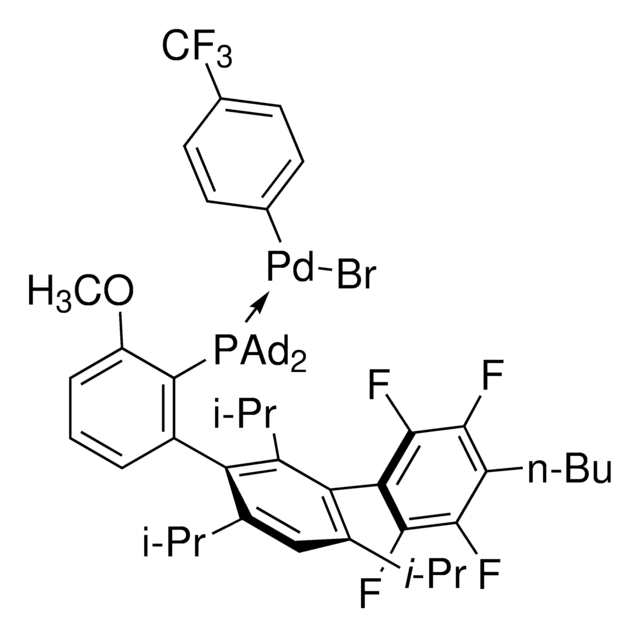
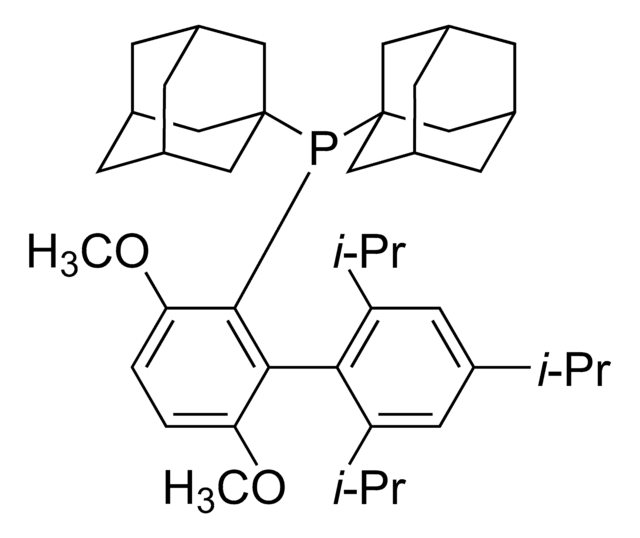
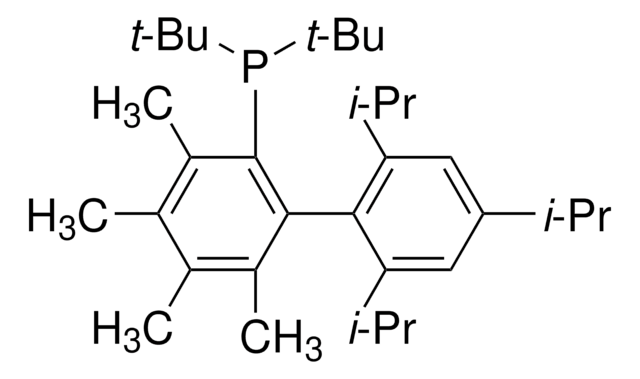
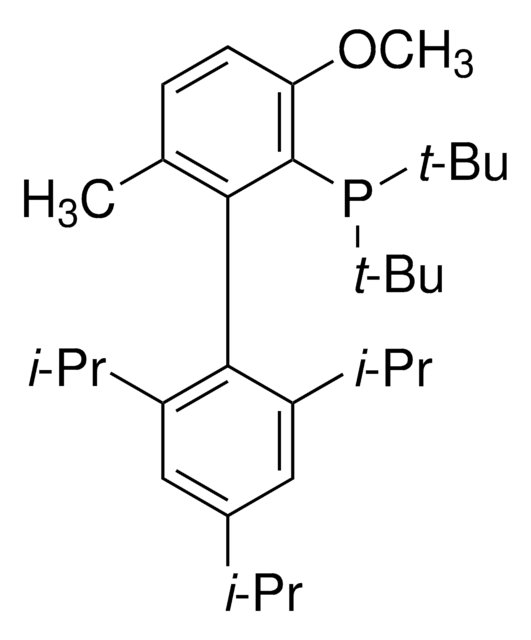
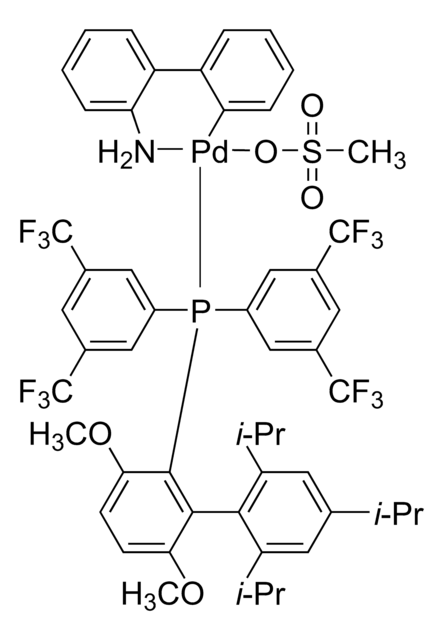
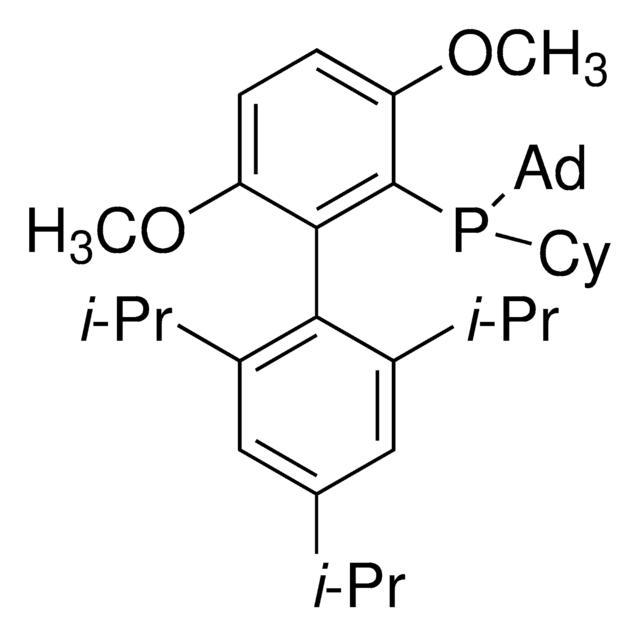
Di-1-adamantyl(4″-butyl-2″,3″,5″,6″-tetrafluoro-2′,4′,6′-triisopropyl-2-methoxy-meta-terphenyl)phosphine
About This Item
Produits recommandés
Pureté
≥95%
Niveau de qualité
Forme
powder
Capacité de réaction
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
Pertinence de la réaction
reagent type: ligand
reaction type: Cross Couplings
Pf
218-223 °C
Groupe fonctionnel
phosphine
Température de stockage
−20°C
InChI
1S/C52H67F4OP/c1-9-10-12-38-46(53)48(55)45(49(56)47(38)54)44-40(29(4)5)21-39(28(2)3)43(42(44)30(6)7)37-13-11-14-41(57-8)50(37)58(51-22-31-15-32(23-51)17-33(16-31)24-51)52-25-34-18-35(26-52)20-36(19-34)27-52/h11,13-14,21,28-36H,9-10,12,15-20,22-27H2,1-8H3/t31-,32+,33?,34-,35+,36?,51+,52?,58?
Clé InChI
ALWIRDZSIXWCBO-VABCSHEKSA-N
Application
- In the Pd-catalyzed Buchwald-Hartwig cross-coupling reactions.
- To synthesize highly regioselective aryl fluorides by Pd-catalyzed fluorination of a variety of activated aryl and heteroaryl triflates and bromides.
- To prepare aryl thioethers by C–S cross-coupling of thiols with aromatic electrophile in the presence of palladium catalyst.
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Fluorine containing aromatics (ArF) are desirable compounds with applications in medicinal chemistry and the agricultural industry.
Fluorine containing aromatics (ArF) are desirable compounds with applications in medicinal chemistry and the agricultural industry.
Fluorine containing aromatics (ArF) are desirable compounds with applications in medicinal chemistry and the agricultural industry.
Fluorine containing aromatics (ArF) are desirable compounds with applications in medicinal chemistry and the agricultural industry.
Contenu apparenté
The Buchwald group has developed a series of highly active and versatile palladium precatalysts and biarylphosphine ligands used in cross-coupling reactions for the formation of C-C, C–N, C–O, C–F, C–CF3, and C–S bonds. The ligands are electron-rich, and highly tunable to provide catalyst systems with a diverse scope, high stability and reactivity. Furthermore, the new series of precatalysts are air-, moisture and thermally-stable and display good solubility in common organic solvents. The use of precatalysts ensures the efficient generation of the active catalytic species and allows one to accurately adjust the ligand:palladium ratio. The ligands, precatalysts and methodology developed in the Buchwald group are user friendly and have rendered previously difficult cross couplings reactions, much easier to achieve.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique