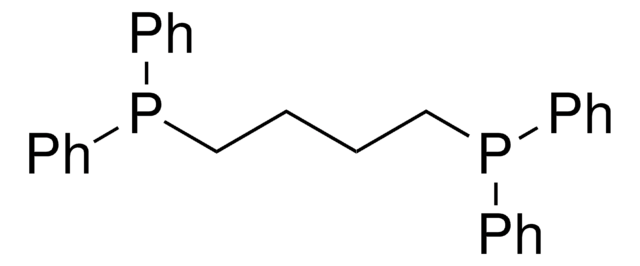
262048
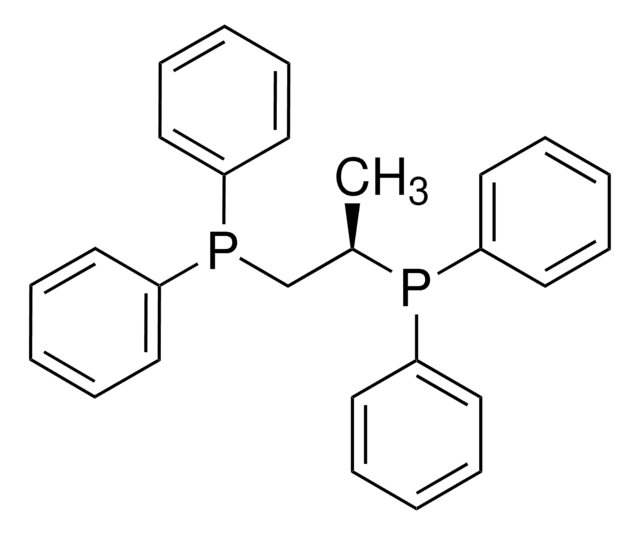
1,3-Bis(diphenylphosphino)propane
97%
Synonyme(s) :
dppp
About This Item
Produits recommandés
Niveau de qualité
Pureté
97%
Forme
solid
Pertinence de la réaction
reagent type: ligand
reaction type: C-C Bond Formation
reagent type: ligand
reaction type: Carbonylations
reagent type: ligand
reaction type: Negishi Coupling
reagent type: ligand
reaction type: Sonogashira Coupling
reagent type: ligand
reaction type: Suzuki-Miyaura Coupling
Pf
63-65 °C (lit.)
Groupe fonctionnel
phosphine
Chaîne SMILES
C(CP(c1ccccc1)c2ccccc2)CP(c3ccccc3)c4ccccc4
InChI
1S/C27H26P2/c1-5-14-24(15-6-1)28(25-16-7-2-8-17-25)22-13-23-29(26-18-9-3-10-19-26)27-20-11-4-12-21-27/h1-12,14-21H,13,22-23H2
Clé InChI
LVEYOSJUKRVCCF-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- Nanoclusterzyme Dual Colorimetric Sensings: The study explored the utility of 1,3-Bis(diphenylphosphino)propane in creating nanoclusterzymes, demonstrating applications in dual colorimetric sensings. This application suggests significant potential in analytical chemistry and sensor development (Zhao et al., 2023).
- Assembly of Icosahedral Units: Research detailed the use of 1,3-Bis(diphenylphosphino)propane in the stronger assembly of icosahedral units within metal clusters, emphasizing its role in enhancing structural stability and potential applications in nanotechnology (Zhou et al., 2023).
- Synthesis and Stability of Gold Clusters: A publication highlighted the synthesis and evaluation of the stability of mixed-diphosphine ligated gold clusters using 1,3-Bis(diphenylphosphino)propane, underlining its critical role in organometallic chemistry and potential industrial applications (Philliber et al., 2022).
- Size-Conversion in Gold Phosphine Clusters: The work investigated core charge density effects on size-conversion from Au(6) P(8) to Au(8) P(8) Cl(2), facilitated by 1,3-Bis(diphenylphosphino)propane, presenting new insights into cluster chemistry and its implications for catalytic and electronic applications (Lv et al., 2020).
- Synthesis of Donor-Acceptor Diblock Copolymer: This study employed 1,3-Bis(diphenylphosphino)propane in sequential polymerizations to synthesize a novel donor-acceptor diblock copolymer, showcasing the versatility of this ligand in polymer chemistry and its potential for creating advanced materials (Ono et al., 2014).
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique





![[1,1′-bis(diphénylphosphino)ferrocène]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)
![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)