Seleccione un Tamaño
| A ustedes/SKU | Disponibilidad | Precio |
|---|---|---|
10 mg | Póngase en contacto con nuestro Servicio de Atención al Cliente para disponibilidad | 125,00 € |
100 mg | Póngase en contacto con nuestro Servicio de Atención al Cliente para disponibilidad | 832,00 € 707,20 € |
Acerca de este artículo
125,00 €
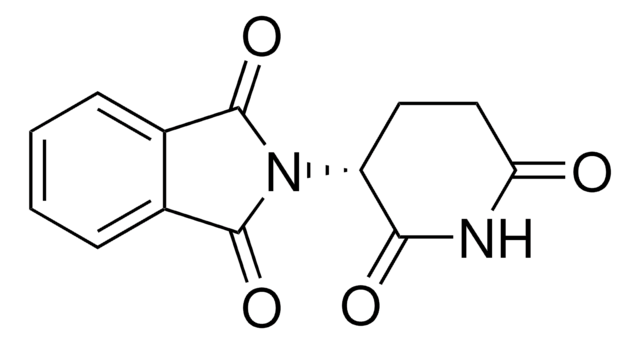
ligand
thalidomide
Quality Segment
assay
≥98% (HPLC)
form
powder
reaction suitability
reagent type: ligand
color
white to beige
solubility
DMSO: 15 mg/mL, clear
originator
Celgene
storage temp.
room temp
SMILES string
O=C1CC[C@@H](N2C(=O)c3ccccc3C2=O)C(=O)N1
InChI
1S/C13H10N2O4/c16-10-6-5-9(11(17)14-10)15-12(18)7-3-1-2-4-8(7)13(15)19/h1-4,9H,5-6H2,(H,14,16,17)/t9-/m1/s1
InChI key
UEJJHQNACJXSKW-SECBINFHSA-N
Gene Information
human ... LITAF(9516), TNF(7124)
mouse ... Nos2(18126)
rat ... Nos1(24598)
Application
Biochem/physiol Actions
Features and Benefits
Preparation Note
1 of 1
Este artículo | |||
|---|---|---|---|
| form powder | form solid | form powder | form powder |
| assay ≥98% (HPLC) | assay >98% | assay ≥98% | assay ≥98% (HPLC) |
| Quality Level 200 | Quality Level - | Quality Level 200 | Quality Level 100 |
| storage temp. room temp | storage temp. - | storage temp. - | storage temp. 2-8°C |
| solubility DMSO: 15 mg/mL, clear | solubility DMSO: soluble, H2O: insoluble, ethanol: insoluble | solubility DMSO: 20 mg/mL, clear | solubility DMSO: ≥14 mg/mL |
| color white to beige | color white | color white | color yellow |
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Repr. 1B
Clase de almacenamiento
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.