Solubility is tested up to 10mg/mL in anhydrous DMSO. Capsazepine should not be reconstituted in aqueous solutions such as water.
Seleccione un Tamaño
| Tamaño de envase | SKU | Disponibilidad | Precio |
|---|---|---|---|
| 5 mg | Póngase en contacto con nuestro Servicio de Atención al Cliente para disponibilidad | 170,00 € | |
| 25 mg | Póngase en contacto con nuestro Servicio de Atención al Cliente para disponibilidad | 575,00 € |
Acerca de este artículo
170,00 €
Nombre del producto
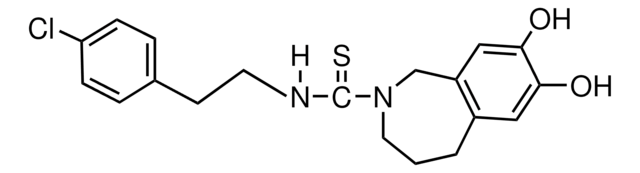
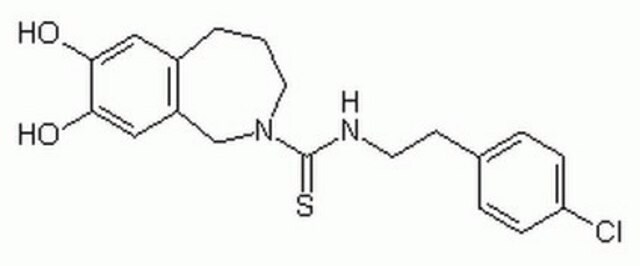
Capsazepine, ≥98% (HPLC), solid
Quality Level
assay
≥98% (HPLC)
form
solid
color
off-white
solubility
DMSO: >10 mg/mL, clear
storage temp.
2-8°C
SMILES string
Oc1cc2CCCN(Cc2cc1O)C(=S)NCCc3ccc(Cl)cc3
InChI
1S/C19H21ClN2O2S/c20-16-5-3-13(4-6-16)7-8-21-19(25)22-9-1-2-14-10-17(23)18(24)11-15(14)12-22/h3-6,10-11,23-24H,1-2,7-9,12H2,(H,21,25)
InChI key
DRCMAZOSEIMCHM-UHFFFAOYSA-N
Gene Information
human ... TRPV1(7442), TRPV4(59341)
rat ... Trpv1(83810)
Application
- to study its effect on capsaicin induced extracellular signal-regulated kinase (ERK) phosphorylation[1]
- to study the role of TRPV1 in central terminals on nociception in rats
- for functional characterization of the TRPV1 in bull spermatozoa[2]
Biochem/physiol Actions
1 of 1
Este artículo | |||
|---|---|---|---|
| form solid | form solid | form powder | form solid |
| assay ≥98% (HPLC) | assay ≥97% (HPLC) | assay ≥98% (HPLC) | assay ≥98% (HPLC) |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| storage temp. 2-8°C | storage temp. 10-30°C | storage temp. 2-8°C | storage temp. 2-8°C |
| solubility DMSO: >10 mg/mL, clear | solubility DMSO: soluble, methanol: soluble | solubility H2O: >10 mg/mL | solubility DMSO: >10 mg/mL |
| color off-white | color off-white | color white | color off-white |
Clase de almacenamiento
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Número de artículo de comercio global
| SKU | GTIN |
|---|---|
| C191-25MG | 04061833468883 |
| C191-5MG | 04061835370177 |
-
With respect to product number catalog C191, concentrations below 10 mg/ml can be diluted with water?
1 respuesta-
¿Le ha resultado útil?
-