155322
trans-Styrylacetic acid
96%
Sinónimos:
4-Phenyl-3-butenoic acid
About This Item
Productos recomendados
Nivel de calidad
Ensayo
96%
puntuación de productos alternativos más sostenibles
old score: 22
new score: 3
Find out more about DOZN™ Scoring
características de los productos alternativos más sostenibles
Atom Economy
Design for Energy Efficiency
Use of Renewable Feedstocks
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
84-86 °C (lit.)
grupo funcional
carboxylic acid
phenyl
categoría alternativa más sostenible
cadena SMILES
OC(=O)C\C=C\c1ccccc1
InChI
1S/C10H10O2/c11-10(12)8-4-7-9-5-2-1-3-6-9/h1-7H,8H2,(H,11,12)/b7-4+
Clave InChI
PSCXFXNEYIHJST-QPJJXVBHSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico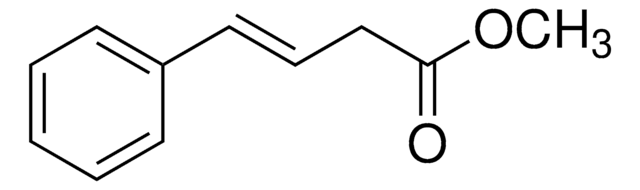



![1,8-Diazabiciclo[5.4.0]undec-7-eno 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)