220201
CHAPS
Molecular Biology Grade
Synonyme(s) :
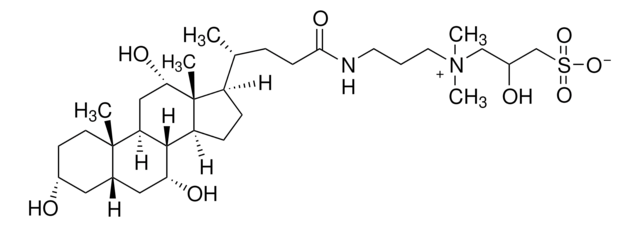
CHAPS, 3-[(3-Cholamidopropyl)dimethylammonio]-1-propanesulfonate
About This Item
Produits recommandés
product name
CHAPS, Molecular Biology Grade,
Qualité
Molecular Biology
Niveau de qualité
Description
zwitterionic
Pureté
≥98% (NMR)
Forme
crystalline powder
Poids mol.
micellar avg mol wt 6150
Fabricant/nom de marque
Calbiochem®
Conditions de stockage
OK to freeze
desiccated (hygroscopic)
Nombre d'agrégation
10
Couleur
white
CMC
6 - 10 mM
6 mM (20-25°C)
Pf
157 °C (315 °F)
Température de transition
cloud point >100 °C
Solubilité
water: 1.0 M
Activité étrangère
DNase, RNase, and Protease, none detected
Conditions d'expédition
ambient
Température de stockage
15-25°C
Chaîne SMILES
C[C@H](CCC(=O)NCCC[N+](C)(C)CCCS([O-])(=O)=O)[C@H]1CC[C@H]2[C@@H]3[C@H](O)CC4C[C@H](O)CC[C@]4(C)[C@H]3C[C@H](O)[C@]12C
InChI
1S/C32H58N2O7S/c1-21(8-11-29(38)33-14-6-15-34(4,5)16-7-17-42(39,40)41)24-9-10-25-30-26(20-28(37)32(24,25)3)31(2)13-12-23(35)18-22(31)19-27(30)36/h21-28,30,35-37H,6-20H2,1-5H3,(H-,33,38,39,40,41)/t21-,22?,23-,24-,25+,26+,27-,28+,30+,31+,32-/m1/s1
Clé InChI
UMCMPZBLKLEWAF-RFCNGIAKSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Description générale
Application
Useful for solubilizing membrane proteins and breaking protein-protein interactions. CHAPS′ small micellar molecular weight (6,150) and high critical micelle concentration (6-10 mM) allow it to be removed from samples by dialysis. It is also suitable for protein solubilization for isoelectric focusing and two-dimensional electrophoresis. CHAPS is commonly used for non-denaturing (without urea) IEF and has been shown to give excellent resolution of some subcellular preparations and plant proteins. Concentrations between 2-4% (w/v) are typically used in an IEF gel.
Avertissement
Reconstitution
Autres remarques
Ofri, D., et al. 1992. J. Neurochem. 58, 628.
Ransom, R.W., et al. 1992. Biochem. Pharmacol. 43, 1823.
Simonds, W.F., et al. 1992. Proc. Natl. Acad. Sci. USA77, 4623.
Yannariello-Brown, J., and Weigel, P.H. 1992. Biochemistry31, 576.
Hjelmeland, L.M. 1980. Proc. Natl. Acad. Sci. USA77, 6368.
Simonds, W.F., et al. 1980. Proc. Natl. Acad. Sci. USA77, 4623.
Informations légales
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 4 Oral
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique![CHAPS (3-[(3-Cholamidopropyl)-dimethylammonio]-propane- sulfonate) for biochemistry](/deepweb/assets/sigmaaldrich/product/structures/265/550/452ffcaa-af89-4e31-99a1-0a0f29072b82/640/452ffcaa-af89-4e31-99a1-0a0f29072b82.png)