804398
[(TMEDA)Ni(o-tolyl)Cl]
95%
About This Item
Produits recommandés
Niveau de qualité
Pureté
95%
Forme
powder
Pertinence de la réaction
core: nickel
reagent type: catalyst
Capacité de réaction
reaction type: Cross Couplings
Chaîne SMILES
CC1=CC=CC=C1[Ni]Cl.CN(C)CCN(C)C
InChI
1S/C7H7.C6H16N2.ClH.Ni/c1-7-5-3-2-4-6-7;1-7(2)5-6-8(3)4;;/h2-5H,1H3;5-6H2,1-4H3;1H;/q;;;+1/p-1
Clé InChI
NMLMESVZRUMFAE-UHFFFAOYSA-M
Catégories apparentées
Application
Produit(s) apparenté(s)
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Palladium catalysts in transition metal-catalyzed coupling reactions offer diversity, accessibility, and robustness over nickel catalysts.
Palladium catalysts in transition metal-catalyzed coupling reactions offer diversity, accessibility, and robustness over nickel catalysts.
Palladium catalysts in transition metal-catalyzed coupling reactions offer diversity, accessibility, and robustness over nickel catalysts.
Palladium catalysts in transition metal-catalyzed coupling reactions offer diversity, accessibility, and robustness over nickel catalysts.
Contenu apparenté
Research in the Doyle group focuses on two areas: nucleophilic fluorination and nickel catalysis. The Doyle group has developed several reagents that advance these research areas. In fluorination, 2-pyridinesulfonyl fluoride (PyFluor) can be used for the mild deoxyfluorination of primary and secondary alcohols, a procedure which is normally accomplished by the sensitive reagent DAST. In nickel catalysis, the Doyle group has developed a modular, air-stable nickel precatalyst, [(TMEDA)Ni(o-tolyl)Cl], which has broad utility for a wide variety of reactions. This precatalyst can be used in place of Ni(cod)2, NiCl2, as well as other reported precatalysts. Doyle has also reported electron-deficient olefin ligands as a new class of ligand for accelerated reductive elimination. In particular, the sultam-derived ligand Fro-DO has been found to be critical for high yields in the cross-coupling of tertiary aziridines to form quaternary centers.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique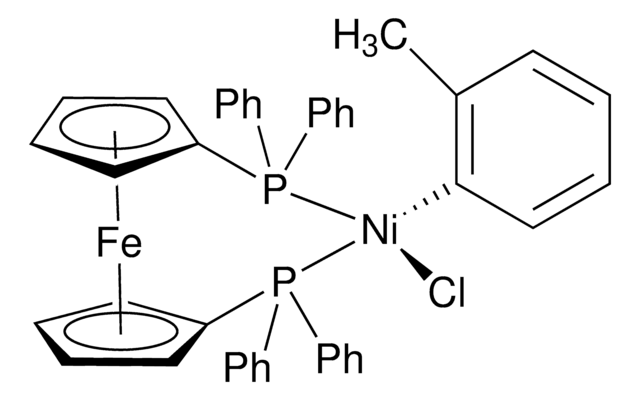

![[(TEEDA)Ni(o-tolyl)Cl] ≥95%](/deepweb/assets/sigmaaldrich/product/structures/156/227/a6ce708d-c671-4ca6-98ba-ef780504ca58/640/a6ce708d-c671-4ca6-98ba-ef780504ca58.png)


![[1,3-Bis(diphenylphosphino)propane]dichloronickel(II)](/deepweb/assets/sigmaaldrich/product/structures/844/065/af07f787-c6a3-4a6e-a22b-47a933c73978/640/af07f787-c6a3-4a6e-a22b-47a933c73978.png)

![[1,2-Bis(diphenylphosphino)ethane]dichloronickel(II)](/deepweb/assets/sigmaaldrich/product/structures/707/956/483e7a6e-5fb5-4e39-abd1-ecf33ccab3cf/640/483e7a6e-5fb5-4e39-abd1-ecf33ccab3cf.png)



![[1,1′-Bis(diphenylphosphino)ferrocene]dichloronickel(II) 97%](/deepweb/assets/sigmaaldrich/product/structures/274/566/a60d6584-163a-4c41-a738-60f8e4d524fa/640/a60d6584-163a-4c41-a738-60f8e4d524fa.png)