521418
4-Cyanophenylboronic acid
≥95%
Synonyme(s) :
(p-Cyanophenyl)boronic acid, 4-Cyanobenzeneboronic acid, 4-Cyanophenylboric acid
About This Item
Produits recommandés
Pureté
≥95%
Pf
>350 °C (lit.)
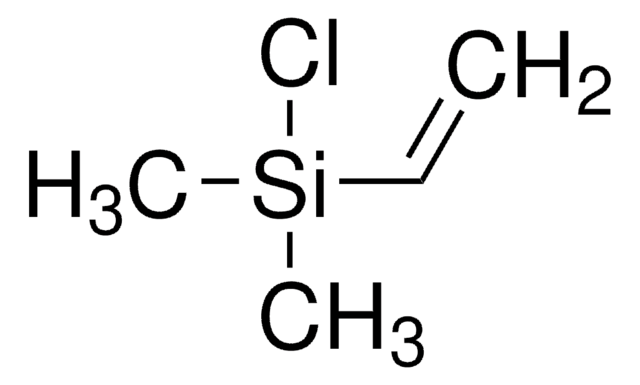
Chaîne SMILES
OB(O)c1ccc(cc1)C#N
InChI
1S/C7H6BNO2/c9-5-6-1-3-7(4-2-6)8(10)11/h1-4,10-11H
Clé InChI
CEBAHYWORUOILU-UHFFFAOYSA-N
Application
- Palladium-catalyzed Suzuki-Miyaura cross-coupling in water.
- Ruthenium catalyzed direct arylation of benzylic sp3 carbons of acyclic amines with arylboronates.
- Ligand-free copper-catalyzed coupling of nitro arenes with arylboronic acids.
- Ferric perchlorate-promoted reaction of fullerenes with various arylboronic acids to give fullerenyl boronic esters.
- Phosphine-free Suzuki-Miyaura cross-coupling.
- Palladacycles as effective catalysts for multicomponent reaction with allylpalladium-intermediates.
- Chan-Lam-type Cu-catalyzed S-arylation of thiols.
- Regioselective cross-coupling reactions under modfied Suzuki and Still cross-coupling reactions with copper catalysis.
- Metal-free biaryl coupling reaction in the presence of dimethyl carbonate as a solvent.
- Suzuki-type cross-coupling reaction with pentavalent triarylantimony diacetates in the absence of a base.
It can also be used to prepare:
- Himbacine analogs as thrombin receptor antagonists and potential antiplatelet agents.
- Trisulfonated calixarene upper-rim sulfonamido and their complexation with trimethyllysine epigenetic mark.
- Antimalarial compounds via Suzuki cross-coupling.
- Deoxyuridine derivatives.
- Oxidative hydroxylation
- Trifluoromethylation
- 1,4-Addition reactions
Precursor in the synthesis of inhibitors such as:
- Tpl2 kinase inhibitors
- P2X7 antagonists used in the treatment of pain
Autres remarques
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique
![[1,1′-bis(diphénylphosphino)ferrocène]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)





![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)