This product is stored at -20°C and is not assigned an expiration or recommended retest date. Products with no expiration date or recommended retest date should be routinely inspected by customers to ensure they perform as expected. These products are also subject to a one-year warranty from the date of shipment. For more information, please access the Product Dating Information document under ADDITIONAL USEFUL DOCUMENTS ABOUT OUR PRODUCTS at the bottom of the Quality Services page with this link: https://www.sigmaaldrich.com/US/en/life-science/quality-and-regulatory-management/quality-services.
Fortfahren mit
Größe auswählen
| Ihnen/SKU | Verfügbarkeit | Preis |
|---|---|---|
5 mg | Ab HEUTE lieferbarvonSchnelldorf | 184,00 € |
25 mg | Ab HEUTE lieferbarvonSchnelldorf | 678,00 € |
Über diesen Artikel
Produktname
MK-571 sodium salt hydrate, ≥95% (HPLC)
Quality Segment
assay
≥95% (HPLC)
form
powder
storage condition
desiccated
color
white to beige
solubility
H2O: 15 mg/mL, clear
originator
Merck & Co., Inc., Kenilworth, NJ, U.S.
shipped in
wet ice
storage temp.
−20°C
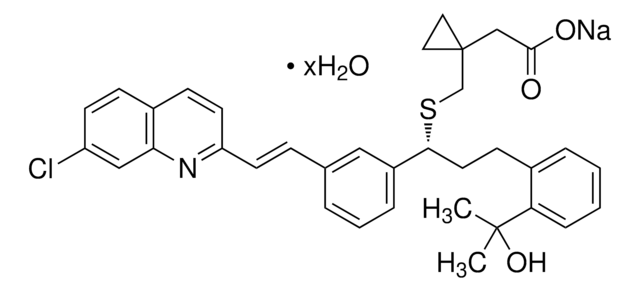
SMILES string
O.[Na+].CN(C)C(=O)CCSC(SCCC([O-])=O)c1cccc(\C=C\c2ccc3ccc(Cl)cc3n2)c1
InChI
1S/C26H27ClN2O3S2.Na.H2O/c1-29(2)24(30)12-14-33-26(34-15-13-25(31)32)20-5-3-4-18(16-20)6-10-22-11-8-19-7-9-21(27)17-23(19)28-22;;/h3-11,16-17,26H,12-15H2,1-2H3,(H,31,32);;1H2/q;+1;/p-1/b10-6+;;
InChI key
MSHRPLRGSQECLY-DOLBFOAYSA-M
Application
- as an efflux inhibitor for monitoring multidrug resistance protein (MRP)-function and to avoid redundancy of other transporters
- to assess its effect on cell proliferation and 2D-migration in vitro in various cell lines of glioblastoma multiforme (GBM)[1]
- as multidrug resistance (MDR) transporter inhibitor to study its effects in ovarian cancer cells
- as specific inhibitors of ABCC1/2 to investigate transport, toxicity, flow cytometry and arsenic efflux
Biochem/physiol Actions
Features and Benefits
1 of 1
Dieser Artikel | |||
|---|---|---|---|
| form powder | form powder | form powder | form solid |
| assay ≥95% (HPLC) | assay ≥98% (HPLC) | assay ≥97% (HPLC) | assay ≥98% (HPLC) |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| storage temp. −20°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
| solubility H2O: 15 mg/mL, clear | solubility DMSO: ≥8 mg/mL at 60 °C | solubility DMSO: ≥5 mg/mL | solubility DMSO: 10 mg/mL, clear (warmed) |
| storage condition desiccated | storage condition desiccated | storage condition - | storage condition desiccated |
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Lagerklasse
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
-
I have a question regarding storage of MK-571 sodium salt hydrate. For how long the powder form can be stored at -20C? Thanks in advance.
1 Antwort-
Hilfreich?
-
-
Does MK-571 sodium salt hydrate has to be prepared freshly before experiments or can it be diluted and stored as a stock solution; without affecting the stability? Thanks in advance
1 Antwort-
Solution stability studies have not been performed on this compound. However, other sources report that the material is stable up tp 6 months when stored in aliquots at -20°C.
Hilfreich?
-