Größe auswählen
90,40 €
492,00 €
Über diesen Artikel
Fortfahren mit
form
powder
mp
210-213 °C (lit.)
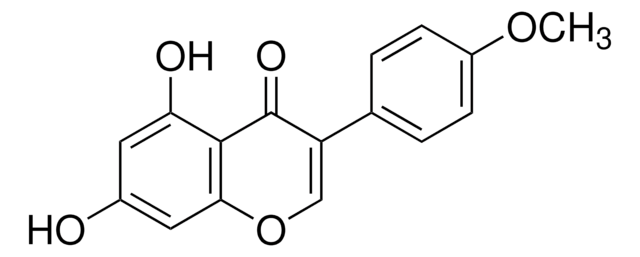
SMILES string
COc1ccc(cc1)C2=COc3cc(O)cc(O)c3C2=O
InChI
1S/C16H12O5/c1-20-11-4-2-9(3-5-11)12-8-21-14-7-10(17)6-13(18)15(14)16(12)19/h2-8,17-18H,1H3
InChI key
WUADCCWRTIWANL-UHFFFAOYSA-N
Gene Information
human ... CYP19A1(1588)
mouse ... Aldh1a2(19378), Maoa(17161)
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
1 of 4
Dieser Artikel | |||
|---|---|---|---|
| form powder | form - | form powder | form - |
| Quality Level 100 | Quality Level 100 | Quality Level - | Quality Level - |
| mp 210-213 °C (lit.) | mp 210-213 °C (lit.) | mp 210-213 °C (lit.) | mp 210-213 °C (lit.) |
| Gene Information human ... CYP19A1(1588) | Gene Information - | Gene Information - | Gene Information - |
General description
Application
Biochem/physiol Actions
Lagerklasse
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Artikel
Antioxidants protect biological systems from oxidative damage produced by oxygen-containing free radicals and from redoxactive transition metal ions such as iron, copper, and cadmium.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| D2016-100MG | 04061833561362 |
| D2016-1G | 04061826016282 |
| D2016-250MG | 04061832407685 |
Aktive Filter
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung