923907
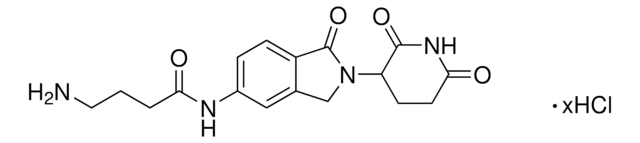
6F,C5-Pomalidomide-piperazine-piperidine-4-carbothioamide hydrochloride
Synonym(e):
2-(2,6-Dioxopiperidin-3-yl)-5-fluoro-6-(4-(piperidine-4-carbonothioyl)piperazin-1-yl)isoindoline-1,3-dione hydrochloride, Crosslinker–E3 ligase ligand conjugate, Protein degrader building block
About This Item
Empfohlene Produkte
ligand
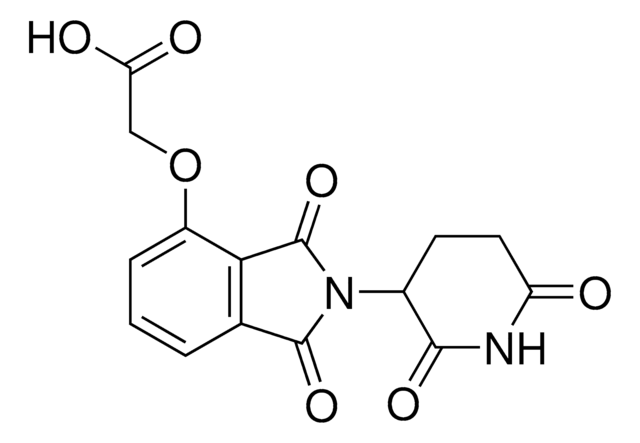
6F,C5-Pomalidomide
Qualitätsniveau
Eignung der Reaktion
reactivity: carboxyl reactive
reagent type: ligand-linker conjugate
Funktionelle Gruppe
amine
Lagertemp.
2-8°C
SMILES String
O=C1C(N2C(C(C=C(F)C(N3CCN(C(C4CCNCC4)=S)CC3)=C5)=C5C2=O)=O)CCC(N1)=O.Cl
Anwendung
Technology Spotlight: Degrader Building Blocks for Targeted Protein Degradation
Protein Degrader Building Blocks
Sonstige Hinweise
Rechtliche Hinweise
Ähnliches Produkt
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Repr. 1B
Lagerklassenschlüssel
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.