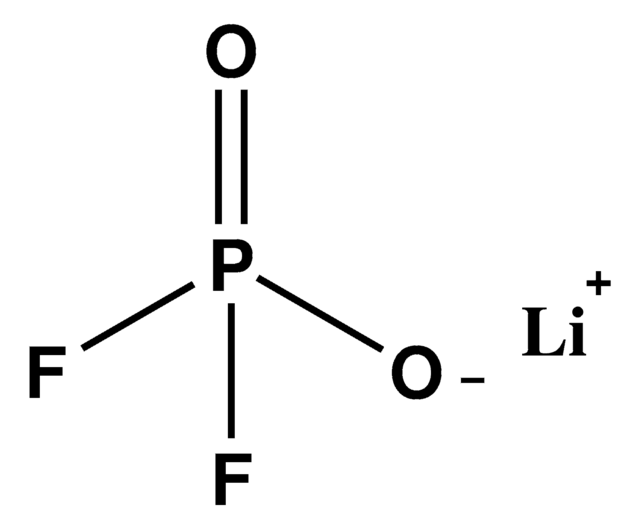
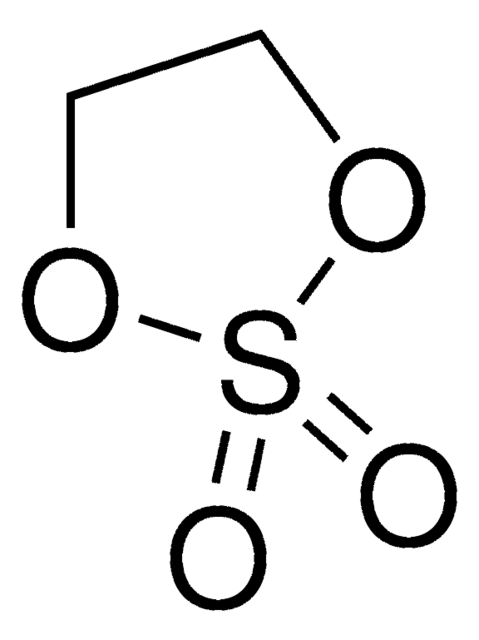
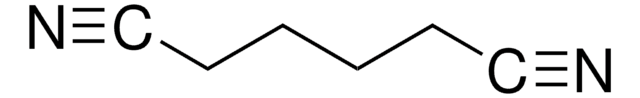
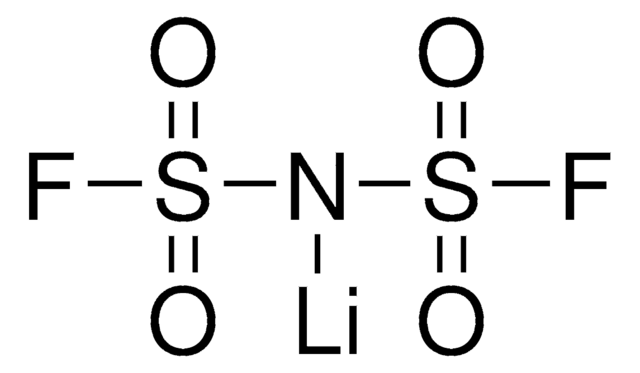774138
Lithium difluoro(oxalato)borate
Synonym(e):
LIDFOB, LIF2OB, LIFOB, LIODFB, Lithium difluoro(ethanedioato)borate, Lithium oxalatodigluoroborate
About This Item
Empfohlene Produkte
Form
powder
Grünere Alternativprodukt-Eigenschaften
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp (Schmelzpunkt)
265-271 °C
Anwendung(en)
battery manufacturing
Grünere Alternativprodukt-Kategorie
, Enabling
SMILES String
F[B-]1(OC(C(O1)=O)=O)F.[Li+]
InChI
1S/C2BF2O4.Li/c4-3(5)8-1(6)2(7)9-3;/q-1;+1
InChIKey
MEDDCIKGDMDORY-UHFFFAOYSA-N
Allgemeine Beschreibung
Anwendung
Leistungsmerkmale und Vorteile
✔ Increases battery life
✔ Stabilizes SEI layer
✔ Suitable for fast charging and low temperatures
Ähnliches Produkt
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Artikel
Solid-state Li batteries: Review of solid electrolytes, ion conduction, structures, and electrochemical processes.
Li-ion batteries are currently the focus of numerous research efforts with applications designed to reduce carbon-based emissions and improve energy storage capabilities.
Lithium-ion batteries offer high energy density and cyclic performance for portable electronic devices.
The critical technical challenges associated with the commercialization of electric vehicle batteries include cost, performance, abuse tolerance, and lifespan.
Verwandter Inhalt
Batteries, fuel cells, and supercapacitors rely on electrochemical energy production. Understand their operation and electron/ion transport separation.
Batterien, Brennstoffzellen und Superkondensatoren sind Energiewandler und Energiespeicher, die auf der elektrochemischen Energieerzeugung an der Grenzfläche zwischen Elektrode und Elektrolyt und der Trennung von Elektronen- und Ionentransport basieren.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.