Alle Fotos(1)
Wichtige Dokumente
424048
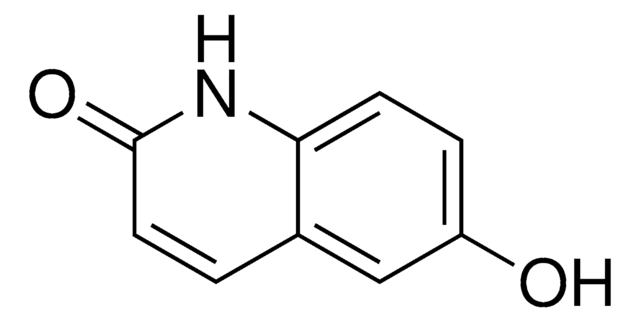
2,6-Chinolindiol
Synonym(e):
2,6-Dihydroxyquinoline, 6-Hydroxycarbostyril
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C9H7NO2
CAS-Nummer:
Molekulargewicht:
161.16
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
mp (Schmelzpunkt)
>300 °C (lit.)
SMILES String
Oc1ccc2nc(O)ccc2c1
InChI
1S/C9H7NO2/c11-7-2-3-8-6(5-7)1-4-9(12)10-8/h1-5,11H,(H,10,12)
InChIKey
AQLYZDRHNHZHIS-UHFFFAOYSA-N
Allgemeine Beschreibung
2,6-Quinolinediol is a substituted quinoline compound. It has been prepared starting from 6-methoxyquinoline. It has been reported to be one of the UV-absorbing compounds (UAC) found in cow milk.
Anwendung
2,6-Quinolinediol may be used as a test compound to investigate the carcinogenicity of the naturally occurring quinoline metabolite of tryptophan. The study was conducted by suspending tryptophan metabolites in cholesterol pellets, followed by the surgical implantation of the pellets into the mice bladder. It may be used as a reactant in the synthesis of 2-chloro-6-quinolinol by reacting with POCl3 (phosphorus oxychloride).
Reactant involved in synthesis of:
- (Benzyloxy)methylquinolinone derivatives for use as PDE3 inhibitors
- Substances related to cilostazol
- 2,6-Quinolinyl derivatives for use as VLA-4 / VCAM-1 antagonists
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
In vivo elution of tryptophan metabolites and other aromatic nitrogen compounds from cholesterol pellets implanted into mouse bladders.
Bryan GT, et al.
Cancer Research, 24(4), 586-595 (1964)
2, 6-Quinolinyl derivatives as potent VLA-4 antagonists.
Lassoie MA, et al.
Bioorganic & Medicinal Chemistry Letters, 17(1), 142-146 (2007)
Mouse bladder carcinogenicity of certain tryptophan metabolites and other aromatic nitrogen compounds suspended in cholesterol.
Bryan GT, et al.
Cancer Research, 24(4), 596-602 (1964)
Pascal Rouge et al.
Food chemistry, 141(3), 1888-1894 (2013-07-23)
The aim of this work was to characterise new UV-absorbing compounds (UAC) in cow milk in order to gain an overview of the molecular diversity of the minor bioactive constituents, that could be used to trace animal feed or that
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.