367796
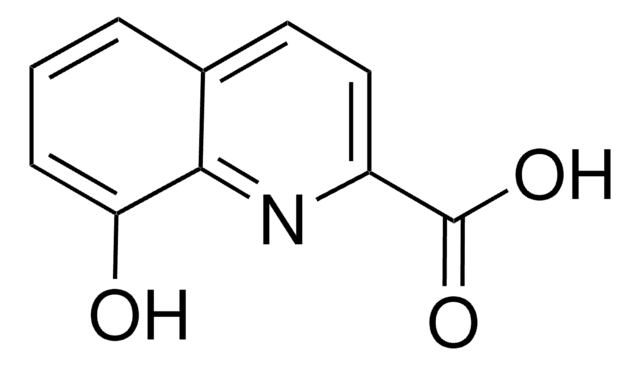
8-Chinolincarbonsäure
98%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C10H7NO2
CAS-Nummer:
Molekulargewicht:
173.17
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
98%
mp (Schmelzpunkt)
183-185 °C (lit.)
Funktionelle Gruppe
carboxylic acid
SMILES String
OC(=O)c1cccc2cccnc12
InChI
1S/C10H7NO2/c12-10(13)8-5-1-3-7-4-2-6-11-9(7)8/h1-6H,(H,12,13)
InChIKey
QRDZFPUVLYEQTA-UHFFFAOYSA-N
Allgemeine Beschreibung
Herbicide 8-quinolinecarboxylic acid and its removal from aqueous solution using sodium montmorillonite, acidic montmorillonite and organo-acidic montmorillonite has been reported.
Anwendung
8-Quinolinecarboxylic acid may be used in the synthesis of:
- novel oxorhenium(V) complexes incorporating quinoline and isoquinoline carboxylic acid derivatives
- chiral 1,2,3,4-tetrahydroquinolinyl-oxazoline compounds, used as ligands for Ru-catalyzed asymmetric transfer hydrogenation of ketones
- chiral quinolinyl-oxazoline compounds, used as ligands for Cu(II) catalyzed asymmetric cyclopropanation
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
S López-Bernabeu et al.
Chemosphere, 144, 982-988 (2015-10-05)
The effect of the electrochemical treatment (potentiostatic treatment in a filter-press electrochemical cell) on the adsorption capacity of an activated carbon cloth (ACC) was analyzed in relation with the removal of 8-quinolinecarboxylic acid pollutant from water. The adsorption capacity of
Chiral quinolinyl-oxazolines as ligands for copper (I)-catalyzed asymmetric cyclopropanation.
Wu X-Y, et al.
Tetrahedron Asymmetry, 9(23), 4143-4150 (1998)
Chiral 1, 2, 3, 4-tetrahydroquinolinyl-oxazoline ligands for Ru-catalyzed asymmetric transfer hydrogenation of ketones.
Zhou Y-B, et al.
Tetrahedron Asymmetry, 13(5), 469-473 (2002)
Barbara Machura et al.
Dalton transactions (Cambridge, England : 2003), 42(24), 8827-8837 (2013-05-04)
Six novel oxorhenium(V) complexes incorporating quinoline and isoquinoline carboxylic acid derivatives were prepared in good yields. Relying on the experimental conditions, compounds with two chelate ligands [ReOCl(iqc)2]·MeOH (1), [ReO(OMe)(iqc)2] (2), [ReO(OMe)(mqc)2] (3) and [ReO(OMe)(8-qc)2] (4) and compounds incorporating one bidentate
M Mekhloufi et al.
Environmental monitoring and assessment, 185(12), 10365-10375 (2013-08-09)
Sodium montmorillonite (Na-M), acidic montmorillonite (H-M), and organo-acidic montmorillonite (Org-H-M) were applied to remove the herbicide 8-quinolinecarboxylic acid (8-QCA). The montmorillonites containing adsorbed 8-QCA were investigated by transmission electron microscopy, FT-IR spectroscopy, X-ray diffraction analysis, X-ray fluorescence thermogravimetric analysis, and
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.