Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 1 mg | Please contact Customer Service for Availability | CZK 5,400.00 |
About This Item
Quality Level
assay
≥98% (HPLC)
form
oil
color
light yellow
solubility
DMSO: freely soluble, ethanol: freely soluble
originator
Bayer
storage temp.
−20°C
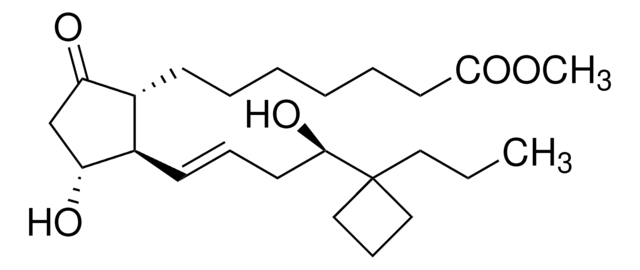
SMILES string
CCCC1(CCC1)[C@H](O)C\C=C\[C@H]2[C@H](O)CC(=O)[C@@H]2CCCCCCC(=O)OC
InChI
1S/C24H40O5/c1-3-14-24(15-9-16-24)22(27)12-8-11-19-18(20(25)17-21(19)26)10-6-4-5-7-13-23(28)29-2/h8,11,18-19,21-22,26-27H,3-7,9-10,12-17H2,1-2H3/b11-8+/t18-,19-,21-,22-/m1/s1
InChI key
XRISENIKJUKIHD-LHQZMKCDSA-N
Application
Biochem/physiol Actions
Features and Benefits
1 of 1
This Item | |||
|---|---|---|---|
| form oil | form oil | form film | form - |
| assay ≥98% (HPLC) | assay ≥98% (HPLC) | assay ≥98% (HPLC) | assay ≥95% (HPLC) |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. −20°C |
| solubility DMSO: freely soluble, ethanol: freely soluble | solubility DMSO: freely soluble, methanol: soluble | solubility DMSO: 10 mg/mL, clear | solubility DMSO: freely soluble, methanol: soluble |
| color light yellow | color colorless to yellow | color colorless | color pale yellow, oil |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Discover Bioactive Small Molecules for Lipid Signaling Research
Related Content
Global Trade Item Number
| SKU | GTIN |
|---|---|
| B6309-1MG | 04061833434598 |
| B6309-5MG | 04061832784335 |